

A MAGIC population as an approach to the conservation and development of genetic diversity of winter barley for breeding purposes by on-farm management
Eine MAGIC-Population als ein Ansatz zur Erhaltung und Entwicklung züchterisch nutzbarer genetischer Diversität von Wintergerste durch on-farm Management
Journal für Kulturpflanzen, 71 (11). S. 286–298, 2019, ISSN 1867-0911, DOI: 10.5073/JfK.2019.11.02, Verlag Eugen Ulmer KG, Stuttgart
 This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/deed.en).
This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/deed.en).Additional genetic diversity in crop breeding programmes and in crop production is required to better cope with the impact of climate change. To complement ongoing activities of winter barley breeders, an evolutionary plant breeding programme has been initiated in 2008. Out of 227 German winter barley varieties released between 1914 and 2003, a set of 58 varieties was genetically analysed using SSR markers. Among these, 32 genotypes representing the genetic diversity of the whole set were crossed according to the Multi-parent Advanced Generation Inter-Cross scheme (MAGIC) over six generations. The resulting 324 lines were combined to form a heterogeneous MAGIC winter barley population. In order to adapt to regional agricultural conditions, MAGIC sub-populations are currently being cultivated within a network of 12 eco-geographically contrasting locations and subjected to natural site-related selection as evolutionary bulks. Cultivation and seed saving will take place over a period of 6 to 8 years under high and low input production conditions. An information system named ROBUSTUM has been developed for consistent recording of cultivation conditions, of characterisation and evaluation data as well as for documentation of the pedigree and for subsequent data analyses. The present approach enables a continuous and dynamic adaptation of the natural genetic diversity present in our crops to climatic and site-related agronomic changes. This makes the evolutionary bulk particularly suitable for organic cultivation, but also for marginal sites and locations with unfavourable soil and weather conditions. In view of increasingly extreme climatic conditions, these properties may become even more important for agriculture in the future. Populations also contribute to the maintenance and development of a genetic diversity as broad as possible. This approach enables estimation of the potential of evolutionary bulks to contribute to the development of sustainable agricultural production systems.
Key words: MAGIC population, evolutionary bulk, winter barley, genetic resources, sustainable agricultural production system
Zusätzliche genetische Diversität in Züchtungsprogrammen und im Wintergerstenanbau sind erforderlich, um den Auswirkungen des Klimawandels besser begegnen zu können. Um bestehende Aktivitäten der Wintergerstenzüchter zu ergänzen, wurde 2008 mit einem Programm zur Populationszüchtung begonnen. Von insgesamt 227 deutschen Wintergerstesorten, die im Zeitraum von 1914 bis 2003 eine Sortenzulassung erlangten, wurden 58 Sorten mit SSR-Markern genetisch analysiert. Davon wurden 32 möglichst divergente Elternlinien ausgewählt und anschließend über 6 Generationen nach einem multiparentalen Kreuzungsschema (MAGIC) miteinander gekreuzt. Die daraus resultierenden 324 Linien wurden zu einer heterogenen MAGIC-Population vereint. Um sich an regionale Bedingungen anzupassen und auszudifferenzieren, werden derzeit MAGIC-Subpopulationen in einem Netzwerk bestehend aus 12 unterschiedlichen, ökogeografisch kontrastierenden Standorten in Deutschland an- und nachgebaut und somit als Evolutionsramsche einer natürlichen standortbedingten Selektion unterzogen. Der Nachbau wird über einen Zeitraum von 6 bis 8 Jahren erfolgen und beinhaltet für jeden Standort zwei unterschiedliche Intensitätsstufen bezüglich Düngung und Pflanzenschutzmaßnahmen. Für die Protokollierung der Anbaubedingungen, die Erfassung von Evaluierungsdaten, aber auch zur Dokumentation des MAGIC-Kreuzungsschemas sowie für nachfolgende Datenanalysen wurde das Informationssystem ROBUSTUM entwickelt. Der beschriebene Ansatz erlaubt eine kontinuierliche und dynamische Anpassung der natürlichen genetischen Vielfalt in unseren Kulturpflanzen an klimatische und standortbedingte agronomische Veränderungen. Damit eignen sich Evolutionsramsche besonders für den ökologischen Anbau, aber auch für Grenzlagen und Standorte mit ungünstigen Boden- und Witterungsbedingungen. Vor dem Hintergrund zunehmender extremer Klimabedingungen können diese Eigenschaften in Zukunft noch eine weitaus größere Bedeutung für die Landwirtschaft erlangen. Populationen tragen ferner zum Erhalt bzw. Aufbau einer möglichst breiten genetischen Vielfalt bei. Mit diesem Ansatz lässt sich das Potenzial von Evolutionsramschen abschätzen, das zur Entwicklung nachhaltiger landwirtschaftlicher Produktionssysteme beitragen kann.
Stichwörter: MAGIC-Population, Evolutionsramsch, Wintergerste, genetische Ressourcen, nachhaltiges landwirtschaftliches Produktionssystem
On-farm management of landraces not only conserves genetic variation but also generates novel variation that enables adaptation of crops to changing environments (Bellon, 2009). Crop landraces formed connected and dynamic networks of genetically diverse populations (Camacho Villa et al., 2005), which were used with the upcoming of science-based plant breeding to develop varieties adapted to modern production systems. In the early phase of modern plant breeding, breeders discarded materials that were not needed to achieve the current breeding targets because such materials could always be obtained from the centers of diversity where numerous landraces were managed by farmers in traditional agricultural production systems. Modern varieties superseded landraces and as a consequence, traditional seed exchange systems were given up and the number of landraces decreased (Kuckuck, 1974, Lopez, 1994, Hammer et al., 1996). Plant breeders expressed concern about the unsustainable treatment of genetic resources already in the first half of the 20th century (Husfeld, 1931) and supported the establishment of national and European programmes to facilitate the collection and long-term conservation of genetic resources in genebanks (IBPGR, 1984). Plant genetic resources programmes were set up and numerous plant exploration missions were organised to rescue and store threatened landraces in genebanks. Later, former breeders’ varieties were also donated to genebanks to allow the recycling of valuable and already adapted material in crop enhancement programmes. Plant genetic resources ensure future breeding progress by providing novel alleles of desired traits. Genetic resources can easily be accessed through genebanks that apply static ex situ management systems to conserve plant genetic resources for food and agriculture. By doing so, genebanks facilitate users’ access to germplasm and related data. Whereas this storage of accessions in genebanks is referred to as ex situ conservation, the on-farm conservation of crop plant germplasm is inter alia referred to as dynamic management of plant genetic resources (Goldringer et al., 2001) and is organised as public-private partnership (Enjalbert et al., 2011).
The pioneers of dynamic management were Harlan and Martini (1929) in California, they started the experimental cultivation of so-called “composite crosses” with barley. From crosses between 28 barley lines, they created a “composite hybrid mixture” to broaden the base of their breeding programme and to increase the chance for selecting improved varieties. The material was grown as unselected bulk for 30 generations to maintain a long-term breeding reserve and achieved the yield level of the best cultivars. Their pioneering work influenced breeding research world-wide (Cooper et al., 2001) and still is doing so. The term “composite cross” was published first by Suneson and Stevens (1953) and describes a population made of progenies derived from several barley varieties that were crossed in all possible combinations or in a similar complex crossing scheme. An evolutionary plant breeding method using such composite crosses was proposed subsequently (Suneson, 1956). A similar project with barley was initiated in Scandinavia. Two bulk populations consisting of 25 adapted varieties, 10 landraces and 5 wild barley populations were produced, crossed several times and cultivated in several places (Veteläinen and Nissilä, 2001).
The development of wheat composite populations was initiated in France in 1984 (Henry et al., 1991) and the material is still reproduced (Enjalbert et al., 2011). The wheat project was planned and sustained by a network of public funded experimental stations over a period of more than 12 years and developed towards a farmers’ network which is a subject of genetic and social studies till these days. The results show that management of genetic resources within a farmers’ network contributes to the long-term conservation of wheat genetic diversity (Thomas et al., 2012) and encouraged us to develop a similar programme for winter barley. This long-term project was coordinated with plant breeding scientists working in the public and private sector and was planned in detail in the year 2007 (Léon et al., 2008).
Modern barley breeding currently relies on genebanks as more or less safe repositories of seed accessions. Unlocking the genetic diversity preserved in genebanks is a prominent task of breeding research. Breeding research supports the development of improved commercial varieties which, when no longer be used, can be donated to genebanks and recycled if need arises (Frese et al., 2007). Evolutionary bulks are artificially created crossing populations that are cultivated under field conditions and adapted locally over generations (Cooper et al., 2001). This does not preserve individual genotypes, but the alleles contained in the bulk. They serve the conservation and development of genetic diversity usable in breeding (Schnell, 1980). The development of multiparent populations can be regarded as active recycling of adapted materials provided by genebanks or made available by breeding companies. The concept of the “renewable genetic resource”, which is described by the terms “dynamic management of plant genetic resources” and “evolutionary plant breeding”, comprises the production of a genetically diverse initial population and its cultivation in ecologically contrasting locations, where it can differentiate genetically over a period of several years under the site-specific selection pressure. Multi-parent Advanced Generation Inter-Cross (MAGIC) populations are created according to the scheme published by Cavanagh et al. (2008) by inter-crossing of several parental founder lines and offspring thereof over several generations until the genetic background of all founders will be equally represented within the inter-crosses. This leads to highly diverse genotypes each with a unique mosaic of founder alleles. The advantage of MAGIC compared to classical biparental populations consists of a higher number of parents and the expected high level of recombination events in the course of inter-crossing results in low linkage disequilibrium (LD) (Stadlmeier et al., 2018). The small number of parents in biparental populations represents only a small portion of the genetic diversity related to the trait of interest. Multi-cross populations were first proposed for plants by Mackay and Powell (2007). MAGIC populations classically are designed to allow for high-resolution mapping of quantitative traits (Sannemann et al., 2015; Huang et al., 2012; Bandillo et al., 2013; Mackay et al., 2014). Furthermore, MAGIC populations can also be used as a long-term genetic resource suitable for plant breeding purposes due to their high heterogeneity (Huang et al., 2015; Ladejobi et al., 2016).
The barley MAGIC population is established to recombine winter barley genetic resources and to adapt the population to diverse regional eco-geographic conditions. As shown for wheat (Enjalbert et al., 2011), the institutional network can develop towards a farmers’ network and help increasing genetic diversity in current production systems as called for by agricultural policy (BMELV, 2007; EC, 2014). In this article, an approach is described that allows for continuous adaptation to changing environmental conditions by natural, site-specific selection of a heterogeneous bulk population. The present approach is comprised of three major parts, firstly the construction of the MAGIC population (2008 until 2015), secondly the cultivation and adaptation within a network of sites (2016 until 2023), and thirdly from 2023 onwards, the overall genetic evaluation of the adapted and differentiated MAGIC population. In addition, the potential of evolutionary bulks contributing to the development of sustainable agricultural production systems will be estimated. In this article, the development of the MAGIC population and the institutional network for winter barley genetic resources are described. The establishment of a network of sites with winter barley populations creates an organisational conservation structure (dynamic management) complementary to the ex situ management as well as develops a research infrastructure component for modern breeding and evolutionary research in cultivated plants (evolutionary bulk winter barley).
Scientific design and construction of the MAGIC population were carried out by the Julius Kühn Institute and was supported by German barley breeders. Suitable founder lines were chosen out of 227 German winter barley varieties released between 1914 and 2003. Based on genetic and phenotypic diversity information from previous studies (Reetz and Léon, 2004; Ordon et al., 2005), a set of 58 varieties was selected. Phillips and Wolfe (2005) proposed the use of molecular marker technologies for the generation of populations and for improving the understanding of the dynamics and genetics of such populations. The selected 58 varieties were therefore genetically analysed using 20 polymorphic SSR markers evenly distributed over the barley genome (Table 1).
Table 1. Markers used to analyse the genetic diversity of 58 winter barley varieties. Repeat motif, chromosomal location and diversity indices (DI) are given. Markers were derived and mapped by Ramsay et al., 2000. nd – not determined
Marker | Repeat motif | Chromosome | Position (cM) | DI |
Bmac0029 | Proprietary markera | 3H | 157 | nd |
Bmac0090 | (AC)20 | 1H | 58 | nd |
Bmac0096 | (AT)6(AC)18 | 5H | 41 | 0.74 |
Bmac0154 | (AT)19(AC)6 | 1H | 84 | nd |
Bmac0156 | (AC)22(AT)5 | 7H | 156 | 0.89 |
Bmac0316 | (AC)19 | 6H | 6 | 0.69 |
Bmag0006 | (AG)17 | 3H | 55 | 0.64 |
Bmag0009 | (AG)13 | 6H | 103 | 0.59 |
Bmag0013 | (CT)21 | 3H | 141 | 0.72 |
Bmag0125 | (AG)19 | 2H | 63 | 0.76 |
Bmag0225 | (AG)26 | 3H | 74 | 0.42 |
Bmag0345 | (CT)6AA(CT)9AA(CT)6AA(CT)7AA(CT)7 | 1H | 66 | 0.29 |
Bmag0359 | (AG)25 | 7H | 100 | 0.52 |
Bmag0749 | (AG)11 | 2H | 151 | 0.64 |
EBmac0701 | (AC)23 | 4H | 76 | 0.74 |
EBmac0970 | (AC)8 | 5H | 54 | 0.49 |
HVM20 | (GA)19 | 1H | 58 | 0.48 |
HVM33 | (CA)7 | 3H | 69 | 0.47 |
HVM67 | (GA)11 | 4H | 118 | 0.53 |
WMC1E8 | (AC)24 | 1H | 164 | 0.43 |
a Bmac0029 is a proprietary marker. Repeat and primer sequences may be obtained from The James Hutton Institute. | ||||
Thirty-two founder lines were identified representing the genetic marker diversity within this set of 58 varieties (Léon et al., 2008) and showing variation in terms of yield, stability, winter hardiness and resistance to leaf rust, net blotch, powdery mildew, BaYMV and BaMMV (Table 2). In the period from 2008 to 2015, the selected winter barley varieties were crossed (Frese et al., 2012) according to the Multi-parent Advanced Generation Inter-Cross scheme (Cavanagh et al., 2008) to produce a heterogeneous multiparent winter barley population.
Table 2. Collection of 32 6-row winter barley varieties used as founder lines.
Variety | Crossing group | Breeding company | Release year | Pedigree |
Sorna | I | Saatzucht Leutewitz | 1989 | ((Maris-Mink × HVW 46172) × Erfa) |
Gaulois | I | Lochow-Petkus GmbH | 1989 | (Gerbel × Athene) |
Vulcan | I | W. von Borries-Eckendorf | 1968 | (Domina × Wong) |
Cervoise | I | Momont Hennette et Fils | 2005 | unknown |
Frances | I | Dippe | 1989 | (Athene × Mammut) |
Peragis | I | Peragis | 1933 | (Giessener-0466 × Friedrichswerther-Berg) |
Birgit | I | W. von Borries-Eckendorf | 1976 | ((Herfordia × H-204) × (70272 × Weissenhaus-38249)) |
Gilberta | I | W. von Borries-Eckendorf | 2001 | (Express × CM-392611) |
Pict | II | KWS UK Limited | 2001 | (92–301 × Musca) |
Alissa | II | Nickerson GmbH | 2000 | (Milva × Krimhild) |
Alinghi | II | Lochow-Petkus GmbH | 2006 | ((LP-6460 × Carola) × Lomerit) |
Merlot | II | Nordsaat Saatzucht GmbH | 2002 | (Theresa × Carola) |
Lomerit | II | Lochow-Petkus GmbH | 2001 | (((Askanova × Grete) × Oceane)) × 133229) |
Leibniz | II | Lochow-Petkus GmbH | 2007 | (LP6–936 × BCYM-5) |
Fridericus | II | Lochow-Petkus GmbH | 2006 | (Carola × LP-6564) |
Naomie | II | Ackermann Saatzucht | 2003 | ((Julia × Nord-51716) × Carola) |
Eiszapfen | III | unknown | unknown | unknown |
Alpaca | III | Semundo Saatzucht GmbH | 1987 | (Banteng × Tapir) |
Banteng | III | Mansholt | 1976 | ((Platen-2349 × Vinesco) × (Dea × Jumbo)) |
Ludmilla | III | Saatzucht Firlbeck GmbH & Co. KG | 1999 | (Hasso × (Banteng × Venus)) |
Mammut | III | W. von Borries-Eckendorf | 1978 | (Vogels.Gold × (Mädru × Weissenhaus 38249)) |
Venus | III | Saatzucht Firlbeck GmbH & Co. KG | 1990 | (Corona × Tapir) |
Engelen II | III | Engelen | 1947 | ((Friedrichswerther Berg × Eckendorfer) × Schwarze) |
Catinka | III | Heidenreich | 1983 | ((Dura × Vogels. Gold) × Mirra) |
Elbany | IV | Lochow-Petkus GmbH | 2002 | ((Borwina × Kantonijo) × P 9147) |
Andrea | IV | W. von Borries-Eckendorf | 1984 | ((Dura × Tocka) × Banteng) |
Engelens-6zlg | IV | Engelen | unknown | unknown |
Hord. Hex. Giganteum | IV | unknown | unknown | unknown |
Vogelsanger Gold | IV | Hauptsaaten für die Rheinprovinz GmbH | 1965 | (((((Isaria × Hord.sp.nigrum-204) × |
Plana | IV | Saatzucht Leutewitz | 1981 | (Vogels.Gold × Klw.1010/60) |
Erfurt | IV | unknown | unknown | unknown |
Traminer | IV | Ackermann Saatzucht | 2002 | (Uschi × Carola) |
Based on a principal component analysis (PCA), 32 diverse founder lines representing the genetic diversity of the 58 analysed varieties were selected and assorted to four groups of eight parents each. The initial crosses were carried out according to this grouping (Table 2). Group I of the parental generation G0 consisted of the pairwise initial crossings (Sorna × Gaulois), (Vulcan × Cervoise), (Frances × Peragis), and (Birgit × Gilberta), group II of (Pict × Alissa), (Alinghi × Merlot), (Lomerit × Leibniz), and (Fridericus × Naomie), group III of (Eiszapfen × Alpaca), (Banteng × Ludmilla), (Mammut × Venus), and (Engelens II × Catinka), and group IV of (Elbany × Andrea), (Engelens-6zlg × Hord. Hex. Giganteum), (Vogelsanger Gold × Plana), and (Erfurt × Traminer). Within each of the four groups, the resulting sixteen G1 lines were intercrossed with offspring resulting from crosses of the same group yielding lines for the next generation G2. To exclude accidental selfings, heterozygosity of G1 and G2 offspring was verified by using selected polymorphic SSR markers. Due to a limited analysis capacity, this validation was restricted to G1 and G2 offspring only. Generally, for each intercrossing generation at least 20 independent plants were used in order to avoid losing alleles. The MAGIC crossing scheme (Cavanagh et al., 2008) was accordingly continued until the production of G6 seeds, each one harboring parts of all genomes from the 32 parental founder lines. Aliquot amounts of seeds from each of the resulting 324 G6 lines were combined in 2015 to form a highly heterogeneous and heterozygous population that was subsequently propagated at the Julius Kühn Institute in Quedlinburg.
For DNA isolation, 3 to 5 cm long leaf pieces from 2-weeks-old barley seedlings were harvested in tubes and the fresh leaf material was vigorously homogenised in a mixer-mill 300 disruptor (Retsch Inc., Hannover, Germany). Genomic DNA was prepared according to the slightly modified CTAB protocol (Saghai-Maroof et al., 1984). The concentration and quality of DNA was determined using a NanoDrop 8000 spectrophotometer (Thermo Scientific, Germany). All samples were adjusted to a final concentration of 30 ng/μl and stored at –20°C in 0.1xTE buffer.
SSR analyses were carried out with selected polymorphic SSR markers (Ramsay et al., 2000). The PCR was performed in a 20 μl reaction volume using the PeqSTAR 96 HPL Thermocycler (PeqLab, Erlangen, Germany). Regarding the primers, two different PCR mixes were used: a) a universal fluorescent-labeled M13-primer (0.07 μM) in combination with an M13-tailed marker- specific forward primer (0.1 μM) and a marker-specific reverse primer (0.17 μM) as described by Schuelke (2000) or b) a Cy5 labeled marker-specific forward primer (0.5 μM) was added to the marker-specific reverse primer (0.5 μM). PCR mix a) was preferred, but in case of unsuccessful amplification, PCR mix b) was employed for the labeling reaction. Generally, a touchdown PCR profile with decreasing annealing temperature by 0.5°C per cycle in the course of 12 cycles from 62°C to 56°C was used that was followed by 35 cycles. However, adapted PCR profiles were applied depending on the specific primer pair employed. The sequence of the forward and reverse primers, the repeat motif and the PCR profiles are given by Ramsay et al. (2000). PCR products were separated and detected on a GenomeLab™ GeXP platform (AB Sciex GmbH, Germany) applying multiplexing.
Each parent variety was genotyped to detect accidental self-pollination and to ensure that only offspring from cross-pollination are used to produce the next generation. Before starting the next crossing step, G1 and G2 offspring was verified with polymorphic SSR markers by means of parent-specific alleles (Table 3). The composite population was finally established by pooling aliquot amounts of 300 seeds of each of the G6 progenies. The bulked seed sample was subsequently propagated in 2015/2016 in Quedlinburg.
Table 3. Polymorphic markers suited to distinguish offspring in the G1 and G2 generation including allele sizes.
G1/G2 | Markera | Parent | specific allele (nt) | Parent | specific allele (nt) |
1 | Bmac0029 | Sorna | 170 | Gaulois | 147 |
2 | GMS001 | Vulcan | 150 | Cervoise | 140 |
3 | Bmag0007 | Pict | 217 | Alissa | 239 |
4 | Bmac0316 | Alinghi | 160 | Merlot | 155 |
5 | Bmag0496 | Frances | 213 | Peragis | 231 |
6 | Bmac0029 | Birgit | 147 | Gilberta | 170 |
7 | Ebmac0788 | Lomerit | 173 | Leibniz | 168 |
8 | Bmac0090 | Fridericus | 223 | Naomie | 225 |
9 | Bmag0007 | Eiszapfen | 215 | Alpaca | 236 |
10 | Bmag0206 | Banteng | 297 | Ludmilla | 273 |
11 | Bmag0206 | Elbany | 273 | Andrea | 297 |
12 | Bmag0225 | Engelens 6 | 159 | H. Hex. Giganteum | (151/159b) |
13 | Bmac0096 | Engelens II | 174 | Catinka | 191 |
14 | Bmac0032 | Mammut | 233 | Venus | 259 |
15 | Bmag0223 | Vogelsanger Gold | 189 | Plana | 173 |
16 | Bmag0140 | Erfurt | 158 | Traminer | 160 |
1 | Bmac0093 | (Sorna × Gaulois) | 161 | (Vulcan × Cervoise) | 157 |
2 | Bmag0206 | (Peragis × Frances) | 273 | (Birgit × Gilberta) | 276 |
3 | Bmag0211 | (Pict × Alissa) | 207 | (Alinghi × Merlot) | 200 |
4 | Bmac0162 | (Lomerit × Leibniz) | 202 | (Naomie × Fridericus) | 212 |
5 | Bmac0209 | (Alpaca × Eiszapfen) | 211 + 229 | (Banteng × Ludmilla) | 209 + 211 |
6 | Bmag0007 | (Engelens II × Catinka) | 213 | (Mammut × Venus) | 217 |
7 | Bmag0013 | (Elbany × Andrea) | 160 | (Engelens 6 × H. Hex. Giganteum) | 158 |
8 | Bmac0032 | (Plana × Vogels. Gold) | 233 | (Traminer × Erfurt) | 239 |
a Detailed information on all used markers was published by Ramsay et al. (2000). | |||||
The advantage of genetically diverse crop populations is mostly perceivable under stress conditions since heterogeneous populations seem to better buffer abiotic and biotic stress effects within the growing season leading to improved yield stability (Schnell and Becker, 1986). Continuous natural as well as artificial selection is likely to lead to populations that perform superior compared to pure stands of the respective parent varieties if reproduced at a specific location for a number of years (Brumlop et al., 2017). Populations therefore constitute an efficient approach to provide heterogeneous material with agronomic superiority particularly under stress conditions (Phillips and Wolfe, 2005). If implemented in a network of eco-geographically contrasting locations, one major advantage of evolutionary population breeding is the high number of possible genotype by environment combinations being represented at the same time. This reduces the likelihood that desirable traits fall through the net during the adaptation process (Döring et al., 2011). However, seed-borne diseases may build up when employing long-term evolutionary breeding because of direct transmission of pathogens to the next generation before adaptation of the population will be effective to limit further increase of disease. In consequence, seed treatments or other conventional crop protection measures have to be taken into consideration to ensure seed quality (Döring et al., 2011).
Fifty-one different soil-climate-areas have been defined for Germany that are derived from meteorological and soil data and result in a consent map of soil-climate-areas released by German agricultural federal and state authorities (Roßberg et al., 2007). Based on these different areas, 12 eco-geographically contrasting locations were selected to cultivate sub-populations in order to promote the development of differently adapted germplasm (Table 4). These locations constitute the network for the dynamic management of winter barley genetic resources and involve three locations under organic management practices, too. In addition, a further location in Southern Sweden near Alnarp was included (Table 4). Since 2016, sub-populations of the material have been continuously cultivated under high and low agricultural input conditions including crop protection measures at each one of these locations. Adaptation of winter barley sub-populations to different climatic, soil and agricultural input conditions will take place over a period of at least 6 to 8 years. It is however expected to maintain the network of sites as a conservation infrastructure for dynamic management of plant genetic resources over a much longer period. In order to maximise adaptation to the different regional conditions, locally usual cultivations practices like tillage, seed bed preparation and seed density are applied at each of the locations. Based on samples taken in each year, changes in the allele frequencies within and between locations will be monitored at the DNA level.
Table 4. Overview of locations growing winter barley sub-populations with corresponding soil-climate-areas, geographical coordinates, management practices and managing institutions.
Location name | Soil-climate-area no. (German description) | Geographical coordinates | Management practice | Managing institution |
Holtsee | 154 (südliches schleswig-holsteinisches Hügelland) | 54.4212 N, 9.8358 E | conventional | Syngenta |
Groß Lüsewitz | 101 (mittlere diluviale Böden MV und Uckermark) | 54.0663 N, 12.3233 E | conventional | Julius Kühn Institute |
Scharnhorst | 146 (Lüneburger Heide/Sandige Böden) | 52.5309 N, 9.5296 E | conventional | Federal Plant Variety Office |
Dahlem | 104 (trocken-warme diluviale Böden des ostdeutschen Tieflandes) | 52.4599 N, 13.2986 E | conventional | Julius Kühn Institute |
Dahnsdorf | 104 (trocken-warme diluviale Böden des ostdeutschen Tieflandes) | 52.1121 N, 12.6358 E | conventional | Julius Kühn Institute |
Quedlinburg | 107 (Lößböden in der Ackerebene Ost) | 51.7705 N, 11.1441 E | conventional | Julius Kühn Institute |
Witzenhausen | 132 (Zentralhessische Ackerbaugebiete, Warburger Börde) | 51.3739 N, 9.9036 E | organic | University of Kassel |
Nossen | 108 (Lößböden in den Übergangslagen Ost) | 51.0534 N, 13.2619 E | conventional | Federal Plant Variety Office |
Dachwig | 107 (Lößböden in der Ackerebene Ost) | 51.0757 N, 10.8618 E | conventional | Federal Plant Variety Office |
Bad Vilbel | 121 (Rheinebene und Nebentäler) | 50.1954 N, 8.7545 E | organic | Dottenfelderhof |
Haßloch | 121 (Rheinebene und Nebentäler) | 49.3778 N, 8.2778 E | conventional | Federal Plant Variety Office |
Freising | 115 (Tertiär-Hügelland Donau-Süd) | 48.4050 N, 11.6941 E | organic | Technical University of Munich |
Alnarp | Götaland\qs southern plains, Swedena | 55.6667 N, 13.1041 E | conventional | Nordic Genetic Resource Centre |
a The German soil-climate-area system is not applicable to this Swedish location | ||||
The winter barley sub-populations are expected to adapt to the specific local conditions. The climatic conditions in Germany have been recorded continuously by meteorological stations in the past. To characterise differences between the locations, 30-year average data on temperature, precipitation, radiation, sunshine, and frost were collected for the period from 1981 to 2010 for each one of the German locations from the ClimateImpactsOnline portal (http://www.climateimpactsonline.com/) at administrative district level. However, climate is steadily changing and the locations may be subjected to different degrees of climate change resulting in the need for local climate change predictions. When designing such a long-lasting project, potential changes of climatic conditions have also to be taken into account. As possible future scenarios, four Representative Concentration Pathways (RCPs) for greenhouse gases were adopted by the United Nations Intergovernmental Panel on Climate Change (IPCC) in its Fifth Assessment Report (IPCC, 2014). These are RCP2.6, RCP4.5, RCP6, and RCP8.5 named according to the radiative forcing in W/m2 in the year 2100 compared to pre-industrial values. RCP8.5 represents the scenario with the highest greenhouse gas emissions and was considered by the IPCC as an extreme case. Nevertheless, current greenhouse gas emission rates match or even exceed the RCP8.5 scenario (Hayhoe et al., 2017), and therefore, this scenario fits the actual development most closely. The Potsdam Institute for Climate Impact Research (PIK) calculated regional climate projections for the next 60 to 100 years by employing the STARS model (STatistical Analogue Resampling Scheme) (Orlowsky et al., 2008) for all German administrative districts. As a result of changing conditions, the climatic water balance will decrease, especially during the main growing season of crops from May to October (Gerstengarbe et al., 2013). This and other meteorological parameters will have implications for future agricultural economy. At the country level, Germany like Northern European countries is supposed to economically benefit from climate change since its current temperature is below the economical optimum (Ricke et al., 2018). At the regional level, PIK used an Integrated Regional Model Assessment to determine future yields of agricultural crops in Germany. Yield decreases are expected for summer-grown crops in these areas of Germany where water availability is limited. On the other hand, winter cereals will be less influenced and may even benefit from the increased precipitation during the winter season (Gerstengarbe et al., 2013). Meteorological and agronomic predictions based on the RCP8.5 scenario and median temperature increase were collected from the ClimateImpactsOnline portal for the period from 2011 to 2040 for each one of the 12 German locations to track and assess future developments of the sub-populations. There are no explicit data on winter barley yields, but winter wheat data can be accessed to infer that most of the German locations may benefit from climate change while some might expect decreased winter cereal yields. The degree of prospected yield changes is remarkable and ranges from 30.9% decrease to 26.7% increase. Details on past and prospected winter wheat yields for the German locations are given in Table 5.
Table 5. Past (years 1981 to 2010), prospected (years 2011 to 2040) average yields of winter wheat, yield fluctuations (in parenthesis) and relative prospected yield changes for the German locations corresponding to the respective administrative districts. Data were collected from ClimateImpactsOnline applying RCP8.5, medium temperature increase. N/A: data not available
Location name | Administrative district | Past yield (t/ha) | Prospected yield (t/ha) | Prospected yield change |
Holtsee | Rendsburg-Eckernförde | 8.1 (1.9 … 10.9) | 5.6 (4.6 … 7.3) | –30.9% |
Groß Lüsewitz | Rostock | 6.0 (2.8 … 8.8) | 7.6 (6.0 … 9.6) | +26.7% |
Scharnhorst | Hannover | 7.0 (4.1 … 10.4) | 7.1 (5.8 … 8.6) | +1.4% |
Dahlem | Berlin | N/A | N/A | N/A |
Dahnsdorf | Potsdam-Mittelmark | 4.8 (2.2 … 8.0) | 4.7 (3.2 … 6.7) | –2.1% |
Quedlinburg | Harz | 6.2 (3.2 … 9.3) | 5.5 (3.5 … 8.7) | –11.3% |
Witzenhausen | Werra-Meißner | 7.0 (4.8 … 9.3) | 7.1 (6.1 … 8.3) | +1.4% |
Nossen | Meißen | 5.8 (3.3 … 8.6) | 5.4 (4.2 … 7.1) | –6.9% |
Dachwig | Gotha | 6.0 (3.2 … 8.8) | 6.8 (5.7 … 8.1) | +13.3% |
Bad Vilbel | Wetteraukreis | 7.0 (4.8 … 9.3) | 7.1 (6.1 … 8.3) | +1.4% |
Haßloch | Bad Dürkheim | 6.0 (3.3 … 8.3) | 6.7 (6.1 … 7.6) | +11.7% |
Freising | Freising | 6.3 (3.2 … 9.1) | 6.4 (5.6 … 7.3) | +1.6% |
In autumn of 2016, all locations were provided with seeds from the propagated winter barley population ERG16–1 and asked to continuously cultivate the population on two plots of approximately 100 m2 applying two different fertilizer and pesticide intensities. Given that the locations correspond to different conditions regarding soil and climate, usual local cultivation practices should be applied in respect to crop rotation, sowing date, sowing density, fertilisation, use of pesticides, and harvest. The intensive or high input variant at each location stands for these usual local cultivation conditions whereas the extensive or low input variant represents reduced fertiliser and pesticide intensities. Low input fertiliser application rate should amount to approximately one third of the high input rate. Regarding the low input variant, plant growth regulators, fungicides and insecticides should be completely avoided and only be used in case of probable total loss of the harvest. Use of pesticides only applies to the conventionally managed locations while locations under organic managing practices completely reject pesticides. Therefore, variants of the organically managed locations only differ in the fertiliser application rate. When needed, special local measures are applied, for example the urban location Dahlem situated in the city of Berlin covers the plots with nets to prevent bird damage after sowing and before harvest. All local treatments and cultivation conditions are being continuously documented. The harvested seeds at each location are saved for sowing the next generation of the population. To avoid possible edge effects, only the harvest of the central part of each plot is kept as seed. The different locations constitute the network of sites since 2016 when sub-populations were first cultivated, and from then, the populations have been grown continuously at the locations. It is intended to continue cultivation and seed saving for 6 to 8 years. In order to ensure this long-term cooperation between the managing institutions of the locations, a memorandum of understanding was signed to promote the continuity of the network. The Julius Kühn Institute was appointed to currently take over the coordination of the network and is in charge of the network management. This includes services like documentation, data management and long-term sample storage.
Samples of approximately one kg of each harvest from all locations are sent every year to the Julius Kühn Institute in Quedlinburg for central storage and later evaluation. For better comparison, evaluation will take place after the period of site-specific adaptation at one location with all the collected samples. Since the adaptation period lasts 6 to 8 years, standard genebank long-term storage measures (Rao et al., 2006) are applied to the seed samples, which includes drying to a low moisture content of about 5% (Ellis and Roberts, 1980) before storage at low temperature (Dickie et al., 1990). Purified and dried seed samples from the locations are stored in air-tight 1000 ml wide-neck transparent PVC containers with PP screw-on caps and PE-foam seals (Carl Roth GmbH + Co. KG, article no. H318.1) at –20°C in a chest freezer. In addition to the samples from all locations and all years, seed samples from the initial evolutionary bulk (ERG16–1) that was sent out to all locations as well as from the 32 parent varieties are treated and stored analogously.
When dealing with populations resulting from multiparent intercrosses, data management and documentation is quite a challenge since their origin is often complex as they may originate from landraces, cultivars, lines, extracts from other populations and the ancestors may be a few or even hundreds of different genotypes obtained through various crossing schemes. Bulk populations have no fixed characteristics and their dynamics depend on the environmental conditions where they have been grown and on population composition, recombination and specific selection methods applied. ROBUSTUM is currently being developed as an information system within a project that aims at inter alia analysing the potential of increased intra-specific diversity within winter wheat populations on their ability to adapt to low input farming conditions in future cropping systems (Baresel et al., 2018). ROBUSTUM was implemented as an information system that on the one hand meets the specific documentation needs of the project but on the other hand exhibits a flexible and extendible structure that makes the system also suitable for other related projects and further documentation purposes (Bülow et al., 2018a). ROBUSTUM is an information system for the consistent documentation of breeding material like varieties and lines, of breeding actions like crossing schemes, to record the composition of populations and derived sub-populations, for documentation of cultivation conditions, characterisation and evaluation data, and for subsequent data analyses. ROBUSTUM is therefore well-suited to record the MAGIC crossing scheme of the present winter barley population as well as for tracing the development of the sub-populations at the different locations. The complex pedigree of the initial winter barley population can be retrieved via ROBUSTUM (Bülow et al., 2018b). For easy access, the system provides an interactive graphical interface for pedigree visualisation (Fig. 1). All steps from the initial crossing of the 32 parental founder varieties, all resulting lines and further crosses through six generations, and the combination of 324 lines to form the bulk population are documented. Additionally, data can be exported in various formats. In the near future, the complete documentation of cultivation conditions at all locations will be transferred to ROBUSTUM. The upcoming evaluation data from the stored samples will be recorded by ROBUSTUM as well.
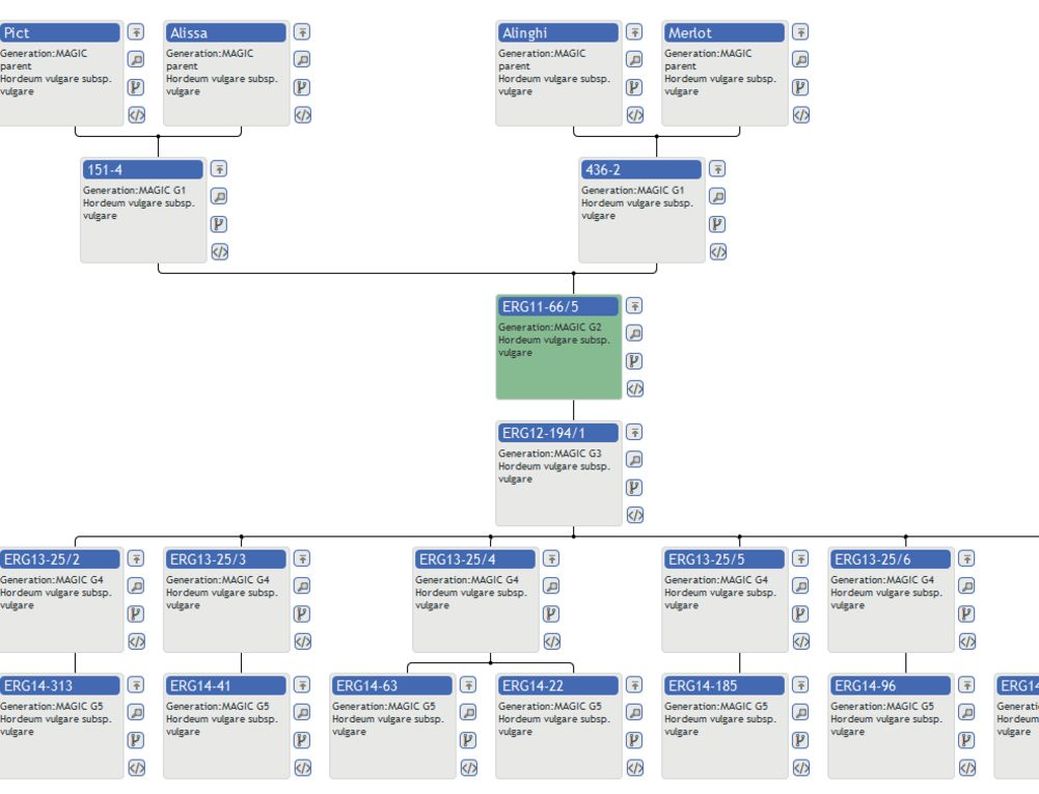
Fig. 1. Pedigree representation by ROBUSTUM for barley line ERG11–66/5 (highlighted) from MAGIC crossing generation G2. The pedigree displays the line’s parentage up to its four parental founder varieties as well as all descendants derived from the line down to generation G5. The number of levels to be shown can be adjusted (not shown) and the interactive functionality enables retrieval of additional details on the breeding material, its pedigree in XML format and the corresponding breeding actions.
Today's elite varieties originated from predominant landraces of the mid-last century, which consisted of a heterogeneous mixture of inbred lines. By targeted selection of individual plants from these populations of landraces, a rapid decline in the genetic variability has been observed. Recent studies revealed a steady reduction of climate resilience in European cereal varieties indicating that breeding efforts so far have not been adequately addressing climatic variability (Kahiluoto et al., 2014; Kahiluoto et al., 2019). Early selection favouring genotypes best-suited for the specific breeding objectives resulted in a strong narrowing of the genetic diversity (genetic bottleneck) which was considered a serious problem in barley breeding (Fischbeck, 1992). Ordon et al. (2005) analysed the temporal changes in genetic diversity in winter and spring barley varieties using 30 SSR markers and noticed a decrease in genetic diversity as measured according to Nei (1973) in winter barley and an increase in spring barley varieties released between 1959 and 2003. The increase of genetic diversity in the analysed spring barley variety collection was interpreted as the result of intensified spring barley breeding since the 1980 s. Genetic bottlenecks are therefore no problem as long as genetic resources are being conserved and sufficient incentives promote the broadening of the breeding pool.
An effective and efficient approach for a dynamic management system with wheat was described by Goldringer et al. (2001) and is being developed by the INRA at Le Moulon (France) towards an on-farm management system involving farmers (Enjalbert et al., 2011). This French approach is being accompanied and supported by a Seed History and Network Management System, an information system called SHiNeMas (Rivière et al., 2013). Farmers in Germany have already expressed interest in the present winter barley population. The network may therefore be broadened towards a future participatory breeding network. ROBUSTUM will take over the documentation required by such a network. Recently, the process to register the winter barley population as a heterogeneous population variety at the German Federal Plant Variety Office has been initiated to allow for easy distribution to interested farmers. The registration of heterogeneous population varieties is also possible in Germany since the publication of the regulation on the placing into the market of seed of populations of the species oat, barley, wheat and corn (BGB, 2015). The regulation refers to the implementing decision of the European Commission on the organisation of a temporary experiment providing for certain derogation rules for the marketing of populations of the plant species wheat, barley, oats and maize (EC, 2014). This experiment is part of the agricultural policy trying to increase inter- und intra specific genetic diversity in crop production as well as on grassland (BMELV, 2007). It is expected that a more diversity-friendly agricultural policy will not only promote sustainable farming activities but also help maintaining and enhancing good ecological infrastructures, and promote the conservation plant genetic resources (Paracchini and Bertaglia, 2018).
The production of and access to food and feed in sufficient quantity and quality today and tomorrow is pivotal to the implementation of the sustainability concept and an enormous challenge to agriculture facing numerous production limits. The challenge to agriculture research today is to investigate and develop land utilisation systems that (i) contribute to the maintenance and improvement of ecosystem function, (ii) treat the non-renewable resources sustainably, (iii) can buffer production systems against negative effects of weather extremes and other hazards, and (iv) can produce food, fodder and other agricultural commodities in sufficient quantities and qualities (Baeumer, 1997; Gregorius, 2001; Fuhrer, 2003). How inter-specific diversity can be deployed to implement resilient production systems is currently being investigated (Lin, 2011; Raseduzzaman and Jensen, 2017). Research projects concern inter alia the effects of intercropping systems on weed control in pea and barley (Corre-Hellou et al., 2011), wheat and subclover (Radicetti et al., 2018), canola and legume mulch (Lorin et al., 2015) or the recovery of fertiliser phosphorus (Xia et al., 2013). Inter-varietal diversity, i.e. specific fractions of the intra-specific diversity combined in varieties of a crop species, offers benefits in yield response, yield stability and disease control (Newton, 2016; Newton and Guy, 2009) in barley. The yield increase or disease index decrease of mixtures compared to the average of their components in pure stands depends on the specific features of the components and how the components interact in the mixture under local production conditions. High intra-specific diversity thus has the potential for environmental resilience, i.e. buffering the effects of environmental and climate variability (Döring et al., 2010). In addition, evolutionary processes contribute to a durable conservation of biodiversity.
Bulk populations constitute a significant contribution to the conservation and development of genetic diversity that can be used in breeding and contribute to the replenishing of the barley gene pool. MAGIC populations compared to classical biparental populations highly benefit from the inter-crossing process due to a faster decay in LD and breakup of initial linkage blocks. The high number of recombination events is also a prerequisite for new allele combinations and influences the genetic diversity of the population. In the case of self-pollinating species, the adaptation and enrichment of favourable alleles can be measured after 6 to 10 generations (Goldringer et al., 2001). Heterozygosity will substantially decrease in the first years after the last crossing and only thereafter, a continuous effect of the selection forces that is not masked by heterozygosity will be detectable. Because of this, in the present barley MAGIC approach cultivation and adaptation within a network of sites shall take place over a long period until 2023. Thereafter, the evaluation of the adapted and differentiated MAGIC sub-populations will be carried out to assess the impact of local adaptation on the allele constitution and to analyse the temporal and local dynamics of biodiversity and the genetic causes of the adaptation processes at molecular level. Specific traits of interest may be identified through marker-trait association studies or high-resolution mapping of quantitative traits by genotyping by sequencing (GBS).
In the future, the MAGIC population may continuously be improved by introducing modern varieties originating from classical plant breeding and use of a male sterility trait to increase recombination rates. This will enable breeders to use the population as a new source for complex traits. The evolutionary plant breeding concept translates the breeding of landraces by farmers and the functions of traditional seed supply systems of the past into science-supported crop diversity management practices of today. Generally, diversification is a usual measure to increase the stability of a system, and even financial service providers diversify their portfolios in particular in periods of increasing instability to minimise the financial risks (Figge, 2001). The present approach addresses the increasing pressure on finite natural resources caused by a growing human population and changing consumption patterns (Steffen et al., 2015) and increasing insecurity in agricultural production systems. Evolutionary plant breeding approaches will also play a very prominent role in solving problems related to climate change (Schimmelpfennig et al., 2018). After successful adaptation, a better fitness of the population against boitic and abiotic stress factors is expected. Thus, the cultivation of evolutionary bulks provides the plant breeders with a continuously filled resource of locally adapted variability that will serve to improve cultivated plants in the future. The creation and cultivation of populations appears to be a suitable measure for successful in-situ conservation of plant genetic resources under on-farm conditions. Bulk populations can be considered as course of action in plant breeding and contribution to a genetic diversification of agricultural production systems.
This project was partly financially supported by the GFPi.
The winter barley MAGIC parents were kindly provided by the department of Plant Breeding of the University of Giessen, INRES Plant Breeding of the University of Bonn and German breeding companies. Thanks to the members of the network for cultivating and harvesting the subpopulations in the past and following years.
Many thanks to Peter Wehling and two anonymous reviewers for revising the manuscript.
The authors have no conflict of interest to declare.
Baeumer, K., 1997: Anbauverhältnis und Fruchtfolge. In: Keller, E.R., Hanus, H., Heyland, K.-U. (eds.) Grundlagen der Pflanzenproduktion. Verlag E. Ulmer, Stuttgart, Germany, 230-233.
Bandillo, N., C. Raghavan, P.A. Muyco, M.A.L. Sevilla, I.T. Lobina, C.J. Dilla-Ermita, C.-W. Tung, S. McCouch, M. Thomson, R. Mauleon, R. Kumar Singh, G. Gregorio, E. Redoña, H. Leung, 2013: Multi-parent advanced generation intercross (MAGIC) populations in rice: progress and potential for genetics research and breeding. Rice 6, 1-15.
Baresel, J.P., G. Backes, J. Baćanović-Šišić, L. Bülow, L. Frese, A.V. Vijaya Bhaskar, M.R. Finckh, S. Knapp, D. Möller, H.J. Reents, J.H. Schmidt, T. Siegmeier, R. Simon, S. Unger, O. Weedon, 2018: INSUSFAR: Innovative approaches to optimize genetic diversity for sustainable farming systems of the future. Inter-specific diversity of wheat for increased resilience and high yields and system specific adaptability. In: Symposium on Breeding for Diversification: A Joint Meeting of the EUCARPIA Section Organic and Low-Input Agriculture, ECO-PB, and the projects LIVESEED, INSUSFAR, DIVERSify, HealthyMinorCereals, ReMIX and Wheatamix. Baćanović-Šišić, J., D. Dennenmoser, M. R. Finckh (eds.), Witzenhausen, Germany, University of Kassel, Kassel University Press, 13-15.
Bellon, M.R., 2009: Do we need crop landraces for the future? Realizing the global option value of in situ conservation. In: Kontoleon, A., U. Pascual, M. Smale (eds.), Agrobiodiversity and Economic Development. Routledge, London and New York, pp. 51-61.
BGB [Bundesgesetzblatt], 2015: Verordnung über das Inverkehrbringen von Saatgut von Populationen der Arten Hafer, Gerste, Weizen und Mais. Teil I, Nr. 32.
BMELV, 2007: Agrobiodiversität erhalten, Potenziale der Land-, Forst- und Fischereiwirtschaft erschließen und nachhaltig nutzen.
Brumlop, S., T. Pfeiffer, M.R. Finckh, 2017: Evolutionary Effects on Morphology and Agronomic Performance of Three Winter Wheat Composite Cross Populations Maintained for Six Years under Organic and Conventional Conditions. Organic Farming 3 (1), 34-50.
Bülow, L., S. Unger, L. Frese, 2018a: Documentation of diversity breeding: The Information system ROBUSTUM. In: Symposium on Breeding for Diversification: A Joint Meeting of the EUCARPIA Section Organic and Low-Input Agriculture, ECO-PB, and the projects LIVESEED, INSUSFAR, DIVERSify, HealthyMinorCereals, ReMIX and Wheatamix. Baćanović-Šišić, J., D. Dennenmoser, M. R. Finckh (eds.), Witzenhausen, Germany, University of Kassel, Kassel University Press, 103-104.
Bülow, L., M. Nachtigall, L. Frese, 2018b: Network for the dynamic management of winter barely genetic resources. In: Symposium on Breeding for Diversification: A Joint Meeting of the EUCARPIA Section Organic and Low-Input Agriculture, ECO-PB, and the projects LIVESEED, INSUSFAR, DIVERSify, HealthyMinorCereals, ReMIX and Wheatamix. Baćanović-Šišić, J., D. Dennenmoser, M. R. Finckh (eds.), Witzenhausen, Germany, University of Kassel, Kassel University Press, 25-26.
Camacho Villa, T.C., N. Maxted, M.A. Scholten, B.V. Ford-Lloyd, 2005: Defining and identifying crop landraces. Plant Genetic Resources 3 (3), 373-384.
Cavanagh, C., M. Morell, I. Mackay, W. Powell, 2008: From mutations to MAGIC: resources for gene discovery, validation and delivery in crop plants. Current Opinion in Plant Biology 11, 215-221.
Cooper, H.D., C. Spillane, T. Hodgkin, 2001: Broadening the genetic base of crops: an overview. In: Broadening the Genetic Base of Crop Production. Cooper, H.D., C. Spillane, T. Hodgkin (eds.), Wallingford, UK, CABI Publishing in cooperation with FAO and IPGRI, CAB International, 1-23.
Corre-Hellou, G., A. Dibet, H. Hauggaard-Nielsen, Y. Crozat, M. Gooding, P.L. Ambus, C. Dahlmann, P. von Fragstein, A. Pristeri, M. Monti, E.S. Jensen, 2011: The competitive ability of pea-barley intercrops against weeds and the interactions with crop productivity and soil N availability. Field Crops Research 122, 264-272.
Dickie, J.B., R.H. Ellis, H.L. Kraak, K. Ryder, P.B. Tompsett, 1990: Temperature and seed storage longevity. Annals of Botany 65,197-204.
Döring, T.F., S. Knapp, G. Kovacs, K. Murphy, M.S. Wolfe, 2011: Evolutionary plant breeding in cereals-into a new era. Sustainability (Switzerland) 3 (10), 1944-1971.
Döring T., M. Wolfe, H. Jones, H. Pearce, J. Zhan, 2010: Breeding for resilience in wheat – Nature’s Choice. In: Breeding for Resilience: a strategy for organic and low-input farming systems? Eucarpia 2nd Conference of the Organic and Low-input Agricultural Section 1-3 December 2010, In: Goldringer I., L. v. B. E. E. Paris, France: 2010, pp.45-48.
EC, 2014: Commission Implementing Decision of 18 March 2014 on the organisation of a temporary experiment providing for certain derogations for the marketing of populations of the plant species wheat, barley, oats and maize pursuant to Council Directive 66/402/EEC (notified under document C(2014) 1681), (Text with EEA relevance), (2014/150/EU).
Ellis, R.H., E.H. Roberts, 1980: The influence of temperature and moisture on seed viability period in barley (Hordeum distichum L.). Annals of Botany 45 (1), 31-37.
Enjalbert, J., J. Dawson, S. Paillard, B. Rhoné, Y. Rousselle, M. Thomas, I. Goldringer, 2011: Dynamic management of crop diversity. From an experimental approach to on-farm conservation. Comptes Rendus Biologies 334, 458-468.
Figge, F., 2001: Biodiversität richtig managen – Effizientes Portfoliomanagement als effektiver Artenschutz. Centre for Sustainability Management e.V., Lüneburg, Germany.
Fischbeck, G., 1992: Barley cultivar development in Europe: success in the past and possible chances in future. In: Barley Genetics VI, Vol. II. Munck, L. (ed.), Copenhagen, Denmark, Munksgaard Int. Publ. Ltd., 885-901.
Frese L., S. Harrer, R. Vögel, 2007: Genetisches Monitoring bei Kulturpflanzen und mit ihnen verwandten Wildarten. In: Monitoring und Indikatoren der Agrobiodiversität, In: Begemann, F., S. Schröder, K.O. Wenkel, H.-J. Weigel (eds.), Tagungsband eines Symposiums am 7. und 8. November 2006 in Königswinter. Agrobiodiversität. Schriftenreihe des Informations- und Koordinationszentrums für Biologische Vielfalt, 66-92.
Frese, L., M. Nachtigall, J. Rode, F. Ordon, J. Léon, 2012: Netzwerk zur Erhaltung der Anpassungsfähigkeit von Kulturpflanzen (NEA*-KULT), Teilvorhaben Wintergerste. Julius-Kühn-Archiv 436, 111-114.
Fuhrer, J., 2003: Agroecosystem Responses to Combinations of Elevated CO2, Ozone, and Global Climate Change. Agriculture, Ecosystems & Environment 97, 1-20.
Gerstengarbe, F.-W., P. Gottschalk, M. Gutsch, F.F. Hattermann, S. Huang, H. Koch, P. Lasch-Born, A. Lüttger, H.J. Schellnhuber, O. Serdeczny, F. Suckow, D. Tiggemann, F. Wechsung, P.C. Werner, 2013: Impacts of climate change for Germany. Potsdam Institute for Climate Impact Research, 2nd edition, August 2013.
Goldringer, I., J. Enjalbert, J. David, S. Paillard, J.L. Pham, P. Brabant, 2001: Dynamic management of genetic resources: a 13-year experiment on wheat. In: Broadening the Genetic Base of Crop Production. Cooper, H. D., C. Spillane, T. Hodgkin (eds.), Wallingford, UK, CABI Publishing in cooperation with FAO and IPGRI, CAB International, 245-260.
Gregorius, H.-R., 2001: Sustainable treatment of resources: The genetic basis. In: Genetic Response of Forest Systems to Changing Environmental Conditions. Müller-Starck G., R. Schubert (eds.) Dordrecht, Netherlands, Kluwer, 203-222.
Hammer, K., H. Knüpffer, L. Xhuveli, P. Perrino, 1996: Estimating genetic erosion in landraces – two case studies. Genetic Resources and Crop Production 43, 329-336.
Harlan, H.V., M.L. Martini, 1929: A composite hybrid mixture. Journal of the American Society of Agronomy and Crop Science Society of America 21, 487-490.
Hayhoe, K., J. Edmonds, R.E. Kopp, A.N. LeGrande, B.M. Sanderson, M.F. Wehner, D.J. Wuebbles, 2017: Climate models, scenarios, and projections. In: Climate Science Special Report: Fourth National Climate Assessment, Volume I. Wuebbles, D.J., D.W. Fahey, K.A. Hibbard, D.J. Dokken, B.C. Stewart, T.K. Maycock (eds.), Washington, DC, USA, U.S. Global Change Research Program, 133-160, doi: 10.7930/J0WH2N54.
Henry, J.P., C. Pontis, J. David, P.H. Gouyon, 1991: An experiment on dynamic conservation of genetic resources with metapopulations. In: Species conservation: a population-biological approach. A. Seitz, V. Loeschcke (eds.), Basel, Schweiz, Birkhäuser Verlag, 185-198.
Huang, B.E., K.L. Verbyla, A.P. Verbyla, C. Raghavan, V.K. Singh, P. Gaur, H. Leung, R.K. Varshney, C.R. Cavanagh, 2015: MAGIC populations in crops: current status and future prospects. Theoretical Applied Genetics 128, 999-1017.
Huang, B.E., A.W. George, K.L. Forrest, A. Kilian, M.J. Hayden, M.K. Morell, C.R. Cavanagh, 2012: A multiparent advanced generation inter-cross population for genetic analysis in wheat. Plant Biotechnology Journal 10,826-839.
Husfeld, B., 1931: Tagung der Internationalen Pflanzenzüchter-Vereinigung in Berlin vom 10. bis 18. Juni 1931. Zeitschrift für Pflanzenzüchtung 14, 697-698.
IBPGR, 1984: The ECP/GR. Introduction to the European Cooperative Programme for Conservation and Exchange of Crop Genetic Resources. AGPG/IBPGR 84/148. UNDP-IBPGR, Rome, Italy.
IPCC, 2014: Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Core Writing Team Pachauri, R.K., L.A. Meyer (eds.), Geneva, Switzerland, IPCC.
Kahiluoto, H., J. Kaseva, J. Balek, J.E. Olesen, M. Ruiz-Ramos, A. Gobin, K.C. Kersebaum, J. Takáč, F. Ruget, R. Ferrise, P. Bezak, G. Capellades, C. Dibari, H. Mäkinen, C. Nendel, D. Ventrella, A. Rodríguez, M. Bindi, M. Trnka, 2019: Decline in climate resilience of European wheat. Proceedings of the National Academy of Science of the United States of America 116 (1), 123-128.
Kahiluoto, H., J. Kaseva, H. Hakala, S.J. Himanen, L. Jauhiainen, R.P. Rötter, T. Salo, M. Trnka, 2014: Cultivating resilience by empirically revealing response diversity. Global Environmental Change 25, 186-193.
Kuckuck, H., 1974: Bedeutung der Nutzung, Erhaltung und Weiterentwicklung der natürlichen genetischen Formenmannigfaltigkeit für die Pflanzenzüchtung – ein Beitrag zur „Grünen Revolution“. Naturwissenschaftliche Rundschau 27, 267-272.
Ladejobi, O., J. Elderfield, K.A. Gardner, R.C. Gaynor, J. Hickey,J.M. Hibberd, I.J. Mackay, A.R. Bentley, 2016: Maximizing the potential of multi-parental crop populations. Applied & Translational Genomics 11, 9-17.
Lin, B.B., 2011: Resilience in agriculture through crop diversification: Adaptive management for environmental change. Bioscience 61, 183-193.
Léon, J., J. Rode, L. Frese, J. Ahlemeyer, F. Ordon, 2008: Aufbau von genetisch diversen Ramschpopulationen für die Wintergerstenzüchtung. In: Mitt. Ges. Pflanzenbauwiss., Band 20 und Vortr. Pflanzenzücht., Band 77. Hoffmann, S., A. Herrmann, F. Taube (eds.), Kiel, Germany, Verlag Schmidt & Klaunig KG, 166-169.
Lopez, P.B., 1994: A new plant disease: uniformity. CERES 26, 41-47.
Lorin, M., M.-H. Jeuffroy, A. Butier, M. Valantin-Morison, 2015: Undersowing winter oilseed rape with frost-sensitive legume livingmulches to improve weed control. European Journal of Agronomy 71, 96-105.
Mackay, I.J., P. Bansept-Basler, T. Barber, A.R. Bentley, J. Cockram, N. Gosman, A.J. Greenland, R. Horsnell, R. Howells, D.M. O’Sullivan, G.A. Rose, P.J. Howell, 2014: An eight-parent multiparent advanced generation inter-cross population for winter-sown wheat: creation, properties, and validation. Genes Genom Genetics 4,1603-1610.
Mackay, I.J., Powell, W., 2007: The significance and relevance of linkage disequilibrium and association mapping in crops. Trends Plant Sci. 12, 53.
Nei, M., 1973: Analysis of Gene Diversity in Subdivided Populations. Proceedings of the National Academy of Science of the United States of America 70 (12), 3321–3323, DOI: 10.1073/pnas.70.12.3321.
Newton, A.C., 2016: Exploitation of Diversity within Crops – the Key to Disease Tolerance? Frontiers in Plant Science 7, 665, DOI: .10.3389/fpls.2016.00665.
Newton, A.C., D.C. Guy, 2009: The effects of uneven, patchy cultivar mixtures on disease control and yield in winter barley. Field Crops Research 110, 225-228.
Ordon, F., J. Ahlemeyer, K. Werner, W. Köhler, W. Friedt, 2005: Molecular assessment of genetic diversity in winter barley and its use in breeding. Euphytica 146, 21-28.
Orlowsky, B., F.W. Gerstengarbe, P.C. Werner, 2008: A resampling scheme for regional climate simulations and its performance compared to a dynamical RCM. Theoretical and Applied Climatology 92, 209-223.
Paracchini, M.L., M. Bertaglia, 2018: Agrobiodiversity and agroecology: state of the art and opportunities in EU policy. Food Security Unit, Land Resources Unit, European Commission, Joint Research Centre.http://www.capsella.eu/wp-content/uploads/2018/05/02_Paracchini_JRC_EC.pdf (Accessed on 12 February, 2019).
Phillips, S.L., M.S. Wolfe, 2005: Evolutionary plant breeding for low input systems. Journal of Agricultural Science 143 (4), 245–254, DOI: 10.1017/S0021859605005009.
Radicetti, E., J.P. Baresel, E.J. El-Haddoury, M.R. Finckh, R. Mancinelli, J.H. Schmidt, I. Thami Alami, S.M. Udupa, M.G.A. van der Heijden, R. Wittwer, E. Campiglia, 2018: Wheat performance with subclover living mulch in different agro-environmental conditions depends on crop management. European Journal of Agronomy 94, 36-45.
Ramsay L., M. Macaulay, S. degli Ivanissevich, K. MacLean, L. Cardle, J. Fuller, K.J. Edwards, S. Tuvesson, M. Morgante, M. Massari, E. Maestri, N. Marmiroli, T. Sjakste, M. Ganal, W. Powell, R. Waugh, 2000: A simple sequence repead-based linkage map of barley. Genetics 156 (4), 1997-2005.
Rao N.K., J. Hanson, M.E. Dulloo, K. Ghosh, D. Nowell, M. Larinde, 2006: Manual of seed handling in genebanks. Handbooks for Genebanks No. 8. Bioversity International, Rome, Italy.
Raseduzzaman, M., E.S. Jensen, 2017: Does intercropping enhance yield stability in arable crop production? A meta-analysis. European Journal Agronomy 91, 25-33.
Reetz, T.C., J. Léon, 2004: Die Erhaltung der genetischen Diversität bei Getreide. Auswahl einer Gersten Core-Collection aufgrund geographischer Herkunft, Abstammung, Morphologie, Qualität, Anbaubedeutung und DNA Markeranalyse. Institut für Pflanzenbau, Professur für Speziellen Pflanzenbau und Pflanzenzüchtung. Forschungsvorhaben im Auftrag des Ministeriums für Umwelt und Naturschutz, Landwirtschaft und Verbraucherschutz des Landes Nordrhein-Westfalen, Bonn, Forschungsbericht Nr. 119, ISSN 1610-2460.
Ricke, K., L. Drouet, K. Caldeira, M. Tavoni, 2018: Country-level social cost of carbon. Nature Climate Change 8, 895-900.
Rivière, P., I. Goldringer, J.F. Berthellot, N. Galic, S. Pin, P. De Kochko, J.C. Dawson, 2013: Response to farmer mass selection in early generation progeny of bread wheat landrace crosses. Renewable Agriculture and Food Systems, 30 (2): 190-201, DOI: 10.1017/S1742170513000343.
Roßberg, D., V. Michel, R. Graf, R. Neukampf, 2007: Definition von Boden-Klima-Räumen für die Bundesrepublik Deutschland. Nachrichtenblatt des Deutschen Pflanzenschutzdienstes 59 (7), 155-161.
Sannemann, W., B.E. Huang, B. Mathew, J. Léon, 2015: Multi-parent advanced generation inter-cross in barley: high-resolution quantitative trait locus mapping for flowering time as a proof of concept. Molecular Breeding 35, 86, DOI: 10.1007/s11032-015-0284-7.
Saghai-Maroof, M.A., K.M. Soliman, R.A. Jorgensen, R.W. Allard, 1984: Ribosomal DNA spacer-length polymorphisms in barley: Mendelian inheritance, chromosomal location, and population dynamics. Proceedings of the National Academy of Science of the United States of America 81, 8014-8018.
Schimmelpfennig, S., C., Heidecke, S. Klages, H. Beer, F. Bittner, S. Krengel, S. Lange (Eds.), 2018: Klimaanpassung in Land- und Forstwirtschaft – Ergebnisse eines Workshops der Ressortforschungsinstitute JKI, FLI, Thünen-Institut. Braunschweig 2018. Thünen Working Paper 86, Johann Heinrich von Thünen-Institut, Braunschweig, Germany, DOI: 10.3220/WP1518167089000.
Schnell, F.W., 1980: Aspekte der genetischen Diversität im Problemkreis der Pflanzenzüchtung. Göttinger Pflanzenzüchter-Seminar 4, 5-15.
Schnell, F.W., H.C. Becker, 1986: Yield and Yield Stability in a Balanced System of Widely Differing Population Structures in Zea Mays L. Plant Breeding 97, 30-38.
Schuelke, M., 2000: An economic method for the Fluorescent labeling of PCR fragments. Nature Biotechnology 18, 233-234.
Stadlmeier, M., L. Hartl, V. Mohler, 2018: Usefulness of a Multiparent Advanced Generation Intercross Population with a greatly reduced mating design for genetic studies in winter wheat. Frontiers in Plant Science 9, 1825.
Steffen, W., K. Richardson, J. Rockström, S.E. Cornell, I. Fetzer, E.M. Bennett, R. Biggs, S.R. Carpenter, W. de Vries, C.A. de Wit, C. Folke, D. Gerten, J. Heinke, G.M. Mace, L.M. Persson, V. Ramanathan, B. Reyers, S. Sörlin, 2015: Planetary boundaries: Guiding human development on a changing planet. Science 347 (6223), DOI: 10.1126/science.1259855.
Suneson, C.A, 1956: An evolutionary plant breeding method. Agronomy Journal 48, 188-191.
Suneson, C.A., H. Stevens, 1953: Studies with bulked hybrid populations of barley. United States Department of Agriculture Technical Bulletin 1067, 1-14.
Thomas, M., E. Demeulenaere, J.C. Dawson, A. Rehman Khan, N. Galic, S. Jouanne-Pin, C. Remoue, C. Bonneuil, I.Goldringer, 2012: On-farm dynamic management of genetic diversity: the impact of seed diffusions and seed saving practices on a population-variety of bread wheat. Evolutionary Applications 5 (8), 779-795.
Veteläinen, M., E.A.J. Nissilä, 2001: Genetic based-broadening of barley in the Nordic Countries. In: Cooper, H. D., C. Spillane, T. Hodgkin (eds.) Broadening the Genetic Base of Crop Production. CABI Publishing, UK.
Xia, H.-Y., Z.-G. Wang, J.-H. Zhao, J.-H. Sun, X.-G. Bao, P. Christie, F.-S. Zhang, L.Li, 2013: Contribution of interspecific interactions and phosphorus application to sustainable and productive intercropping systems. Field Crops Research 154, 53-64.