

Nickel toxicity in Brassica rapa seedlings: Impact on sulfur metabolism and mineral nutrient content
Nickeltoxizität bei Brassica rapa Sämlingen: Einfluss auf den Schwefelstoffwechsel und den Mineralstoffgehalt
Journal für Kulturpflanzen, 72 (9). S. 473–478, 2020, ISSN 1867-0911, DOI: 10.5073/JfK.2020.09.03, Verlag Eugen Ulmer KG, Stuttgart
 | This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/deed.en). Dies ist ein Open-Access-Artikel, der unter den Bedingungen der Creative Commons Namensnennung 4.0 International Lizenz (CC BY 4.0) zur Verfügung gestellt wird (https://creativecommons.org/licenses/by/4.0/deed.de). |
Throughout the world anthropogenic activity has resulted in enhanced soil nickel (Ni2+) levels, which may negatively affect plant productivity. The physiological background of Ni2+ phytotoxicity is still largely unclear. Ten-day exposures of Brassica rapa seedlings to 1, 2 and 5 μM NiCl2 resulted in strongly enhanced tissue Ni levels, a decreased biomass production and leaf chlorosis at ≥ 2 μM Ni2+. At 5 μM Ni2+ plant growth was completely halted. Ni toxicity occurred when the content of the shoot exceeded 1.0 μmol g–1 dry weight and that of the root, 23 μmol g–1 dry weight. Ni2+ exposure at ≤ 2 μM only slightly affected the mineral nutrient content of both shoot and root. Hence, Ni2+ exposure hardly affected the sulfur metabolite content of the plant. At ≥ 1 μM Ni2+ the total sulfur content of the root was only slightly lowered, which could fully be ascribed to a decreased sulfate content. Moreover, the water-soluble non-protein thiol content of both shoot and root was only enhanced at 5 μM Ni2+. From these results it was clear that sulfur metabolism was unlikely to be directly involved in the Ni2+ tolerance mechanisms of B. rapa.
Key words: toxic metals; heavy metals; nickel; sulfur; thiols; glutathione; mineral composition
Weltweit haben anthropogene Aktivitäten zu erhöhten Nickelgehalten im Boden (Ni2+) geführt, was sich negativ auf die Pflanzenproduktivität auswirken kann. Der physiologische Hintergrund der Ni2+ Phytotoxizität ist noch weitgehend unklar. Eine zehntägige Exposition von Brassica rapa Sämlingen mit 1, 2 und 5 μM NiCl2 führte zu stark erhöhten Ni Gehalten im Gewebe, einer verringerten Biomasseproduktion und zu Blattchlorosen bei Konzentrationen von ≥ 2 μM Ni2+. Bei einer Konzentration von 5 μM Ni2+ war kein Pflanzenwachstum mehr zu beobachten. Eine Ni Toxizität trat auf, wenn der Ni Gehalt im Sprosses 1,0 μmol g–1 Trockengewicht und der in der Wurzel 23 μmol g–1 Trockengewicht überschritt. Eine Ni2+ Exposition von 2 μM beeinflusste den Mineralstoffgehalt in Spross und Wurzel nur geringfügig. Daher beeinflusste eine Ni2+ Exposition die Gehalte an Schwefelmetaboliten in der Pflanze kaum. Bei ≥ 1 μM Ni2+ war der Gesamtschwefelgehalt der Wurzel nur geringfügig erniedrigt, was vollständig auf einen verminderten Sulfatgehalt zurückzuführen war. Darüber hinaus war der Gehalt an wasserlöslichen Nicht-Protein-Thiolen sowohl im Spross als auch in der Wurzel nur bei 5 μM Ni2+ erhöht. Aus diesen Ergebnissen geht hervor, dass der Schwefelstoffwechsel wahrscheinlich nicht direkt an den Ni2+ Toleranzmechanismen von B. rapa beteiligt ist.
Stichwörter: Toxische Metalle; Schwermetalle; Nickel; Schwefel; Thiole; Glutathion; Mineralstoffzusammensetzung
Nickel (Ni) is considered as an important micronutrient for the physiological functioning of plants (Wood et al., 2004; Brown, 2006; Polacco et al., 2013; Shahzad et al., 2018). Plants acquire Ni in the divalent form (Ni2+), which is both passively and actively taken up by the root (Cataldo et al., 1988). After uptake, Ni2+ is complexed with organic acids and amino acids, which may subsequently be transported from the root to the shoot or stored in the vacuole (Cataldo et al., 1988).
Ni is required for the activation of ureases, which catalyze the conversion of urea into ammonium (Polacco, 1977; Polacco et al., 2013). Consequently, if plants are grown with urea as sole nitrogen source, Ni deprivation may retard growth by inducing nitrogen deficiency (Polacco, 1977; Polacco et al., 2013). By contrast, if plants are supplied with other nitrogen sources, Ni-deprivation may retard growth by strongly enhancing tissue urea levels (Gerendas et al., 1999; Brown, 2006; Shahzad et al., 2018). Apart from activating ureases, Ni may also be involved in the activation of other enzymes. For instance, it may activate glyoxalase I, which functions in the degradation of methylglyoxal, a toxic molecule produced during cellular processes (Fabiano et al., 2015). Ni may also be transported from plant cells to plant-associated microbes (Brown, 2006). Nitrogen-fixing symbionts and leaf commensals contain Ni-dependent hydrogenases, whose activity is inhibited upon colonization of Ni-deficient plants (Holland & Polacco, 1992).
Despite its significance in plant functioning, elevated Ni levels in the root environment may cause growth retardations and leaf chlorosis (Freeman et al., 2004; Brown, 2006; Shahzad et al., 2018). Elevated soil Ni levels may be the consequence of anthropogenic activities, which include mining, smelting, waste disposal and industrial undertakings where Ni is used as catalyst (e.g., the production of electrical batteries; Brown, 2006; Shahzad et al., 2018). In polluted regions soil nickel levels have increased up to 20 to 30-fold (up to 26 g kg–1) compared to unpolluted regions, which is now seriously threatening agricultural productivity (Brown, 2006; Shahzad et al., 2018).
The primary cause of Ni phytotoxicity remains poorly understood. Analogous to other heavy metals, Ni might react with thiol moieties present in enzymes and other proteins (Pilon et al., 2009; Yadav, 2010). Additionally, exposure to excessive Ni could possibly hamper the root uptake of other essential mineral nutrients (Pilon et al., 2009; Yadav, 2010). Consequently, it may disturb metabolism, which may lead to the production of reactive oxygen species and subsequently lipid peroxidation, protein denaturation and DNA mutation reactions (Pilon et al., 2009; Yadav, 2010).
To tolerate elevated Ni levels, plants could sequester Ni in the vacuole, synthesize Ni-chelator complexes in the cytosol or increase antioxidant levels (Brown, 2006; Shahzad et al., 2018). Sulfur metabolism may have significance in these Ni tolerance strategies. The thiol groups of cysteine and phytochelatins have the chemical properties to complex with heavy metals (Freeman et al., 2004; Sirko & Gotor, 2007; Cuypers et al., 2009). Furthermore, glutathione has the capability to bind reactive oxygen species, which presence might be induced in plants upon exposure to heavy metal stress (Freeman et al., 2004; Sirko & Gotor, 2007; Cuypers et al., 2009). Nevertheless, the physiological significance of these non-protein thiols for Ni tolerance remains elusive. Therefore, the current research describes Ni toxicity and its impact on S metabolism and mineral nutrient content in Brassica rapa seedlings.
Seeds of Brassica rapa cv. Komatsuna (Nickerson-Zwaan, Made, The Netherlands) were germinated in vermiculite in a climate-controlled room. After 10 days, seedlings were transferred to an aerated 25% Hoagland nutrient solution (see Koralewska et al., 2007 for composition) containing 0, 1, 2 or 5 μM NiCl2·4 H2O in 30 l plastic containers (10 sets of plants per container, 3 plants per set). Day and night temperatures were 21 and 18°C (± 1°C), respectively, relative humidity was 70–80% and the photoperiod was 14 h at a photon fluence rate of 400 ± 30 μmol m–2 s–1 (within the 400–700 nm range) at plant height, supplied by Philips GreenPower LED (red/white 120) production modules. After 10 days of exposure, plants were harvested 3 h after the onset of the light period. The roots were rinsed in ice-cold de-mineralized water (3 × 20 s). Subsequently, shoots and roots were separated and weighted. Plant biomass production was calculated by subtracting the initial, pre-exposure, weight from that at harvest. For the determination of dry matter content, plant material was dried at 80°C for 24 h. Four independent experiments were performed for the analyses of the different parameters.
Chlorophyll was extracted from shoots, which were stored at –80°C after harvest, by homogenization in 96% ethanol using an Ultra Turrax (10 ml g–1 fresh weight). After centrifugation at 800 g for 20 min, the chlorophyll content was determined according to Lichtenthaler (1987). Chlorophyll a fluorescence was measured on the adaxial side of fully-expanded leaves as described by Shahbaz et al. (2010a) using a modulated fluorometer (PAM 2000, Walz GmbH, Effeltrich, Germany). For the analyses of Ni, S and other mineral nutrients dried whole shoots and roots were pulverized with a Retsch MixerMill (type MM2; Haan, Germany), digested with nitric acid/perchloric acid and analyzed by inductively coupled plasma optical emission spectroscopy (ICP-OES) as described by Reich et al. (2017). Sulfate was extracted from frozen plant material and determined refractrometrically after HPLC separation (Shahbaz et al., 2010a). Water-soluble non-protein thiols were extracted from fresh shoots and roots (Shahbaz et al., 2010a) and determined colorimetrically after reaction with 5,5’-dithiobis(2-nitrobenzoic acid), according to De Kok et al. (1988).
Statistical analyses were performed using GraphPad Prism (GraphPad Software, San Diego, CA, USA). To compare treatment means a one-way ANOVA with a Tukey’s HSD test as post-hoc test at the P ≤ 0.05 level was performed.
Seedlings of B. rapa were highly susceptible to elevated Ni2+ levels in the root environment, since already at 2 μM Ni2+ the plant biomass production was reduced by 35% (Fig. 1). At 2 μM Ni2+ shoot biomass production was more affected than root biomass production, since the shoot/root ratio was reduced by 21% (Fig. 1). 5 μM Ni2+ completely halted plant growth and it reduced biomass production by 100% (Fig. 1). Ni2+ exposure hardly affected the dry matter content of the seedlings. The dry matter content of the shoot was only slightly enhanced at 5 μM Ni2+, whereas that of the root was not affected by Ni2+ exposure (Fig. 1). A similarly high Ni2+ susceptibility was observed in e.g., soybean (Glycine max; Reis et al., 2017). Moreover, the susceptibility of B. rapa to Ni2+ was comparable with the susceptibility of Brassica species to elevated Zn2+ and Cu2+ levels in the root environment, which also substantially reduced plant growth at concentrations ≥ 2 μM (Shahbaz et al., 2010a; Stuiver et al., 2014). Nevertheless, the Ni2+ susceptibility of Brassica was much higher than that for Mn2+ and MoO42-, which only reduced plant growth at concentrations ≥ 20 and 100 μM, respectively (Neves et al., 2017; Zuidersma et al., 2020). Exposure of B. rapa to Ni2+ inhibited the shoot biomass production more than that of the root (Fig. 1). Exposure of Brassica species to toxic Zn2+ and Mn2+ levels also mainly inhibited shoot growth, whereas exposure to Cu2+ and MoO42- mainly inhibited root growth (Shahbaz et al., 2010a; Stuiver et al., 2014; Zuidersma et al., 2020).
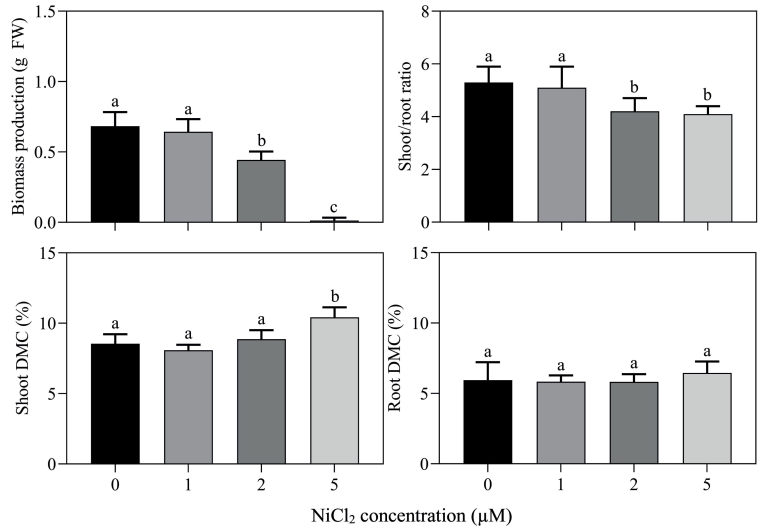
Fig. 1. Impact of Ni2+ exposure on the growth of Brassica rapa. 10-day old seedlings were grown on 25% Hoagland nutrient solutions containing supplemental NiCl2 levels of 0, 1, 2 or 5 μM for 10 days. The initial plant weight was 0.178 ± 0.007 g. Data on biomass production (g FW) and shoot/root ratio represent the mean of four independent experiments with nine to ten measurements with three plants in each (± SD). Data on dry matter content (DMC; %) represent the mean of three independent experiments with three measurements with three plants in each (± SD). Different letters indicate significant differences between treatments (P ≤ 0.05, one-way ANOVA; Tukey’s HSD test as a post-hoc test).
Upon Ni2+ exposure of B. rapa and Cu2+, MoO42-, Ni2+ and Zn2+ exposure of other Brassica species, the decrease in biomass production was accompanied or even preceded by a decrease in the chlorophyll content (Table 1; Shahbaz et al., 2010a; Stuiver et al., 2014; Neves et al., 2017; Zuidersma et al., 2020). At 5 μM Ni2+ the chlorophyll content was lowered by 53% (Table 1). However, the chlorophyll a/b ratio and chlorophyll a fluorescence (Fv/Fm) were not substantially affected by exposure of Brassica to Ni2+ and other heavy metals (Table 1; Shahbaz et al., 2010a; Stuiver et al., 2014; Neves et al., 2017; Zuidersma et al., 2020). The chlorophyll a fluorescence was only slightly decreased upon exposure to 5 μM Ni2+ (Table 1). Apparently, heavy metal exposure of Brassica hampers the development of new chloroplasts, but not the functioning of the already existing chloroplasts (Shahbaz et al., 2010a; Stuiver et al., 2014; Neves et al., 2017; Zuidersma et al., 2020). Thus, the photosystems of the remaining chloroplasts are not or only minimally suffering from photo-inhibition.
Table 1. Impact of Ni2+ exposure on the chlorophyll content and fluorescence of Brasica rapa. 10-day old seedlings were grown on 25% Hoagland nutrient solutions containing supplemental NiCl2 levels of 0, 1, 2 or 5 μM for 10 days. Data on chlorophyll content (mg g–1 FW) and ratio represent the mean of two independent experiments with three measurements with three plants in each (± SD). Data on chlorophyll a fluorescence (Fv/Fm) represent the mean of ten measurements on different plants (± SD). Different letters indicate significant differences between treatments (P ≤ 0.05, one-way ANOVA; Tukey’s HSD test as a post-hoc test).
| NiCl2 concentration (μM) | |||
| 0 | 1 | 2 | 5 |
Shoot |
|
|
|
|
Chlorophyll | 0.70 ± 0.12a | 0.70 ± 0.06a | 0.64 ± 0.04a | 0.33 ± 0.06b |
Chlorophyll a/b | 3.0 ± 0.3a | 3.0 ± 0.2a | 2.7 ± 0.3a | 3.5 ± 1.1a |
Fv/Fm | 0.84 ± 0.02a | 0.83 ± 0.01ab | 0.80 ± 0.01b | 0.74 ± 0.05c |
Exposure of B. rapa to Ni2+ resulted in a strongly enhanced Ni content of the shoot and root (Table 2). However, Ni contents increased more in the root than in the shoot and at toxic Ni2+ levels (2 μM) its content in the shoot increased 34-fold (to 1.04 μmol g–1 dry weight) and in the root 450-fold (to 22.5 μmol g–1 dry weight; Table 2). It has been suggested that excessive Ni2+ and other heavy metal levels in the root environment might be phytotoxic, because their uptake might potentially hamper the uptake of other essential metal ions (Pilon et al., 2009; Yadav, 2010). However, it is doubtful if an altered essential metal ion uptake is the direct cause of Ni2+ toxicity in B. rapa. Exposure of plants to toxic Ni2+ levels (2 μM) only slightly affected the content of other essential mineral nutrients in the shoot and root (Table 2). There was a 30% decrease in the K content of the shoot and a 20 and 50% increase in the Mn and Fe content of roots, respectively, though this also occurred upon exposure to 1 μM Ni2+ (viz. a non-toxic Ni2+ concentration). At 5 μM Ni2+, where biomass production was complete halted, there was a much more pronounced impact on the plant’s mineral nutrient content, with alterations in the root and shoot Cu, Fe, K, Mg, Mn, S and Zn contents (Table 2). In accordance with these results, exposure of Brassica to excessive Zn2+, Cu2+, Mn2+ as well as MoO42- also hardly affected the tissue content of mineral nutrients, suggesting that in Brassica an altered mineral nutrient uptake is unlikely to be the direct cause of toxicity of these metals (Shahbaz et al., 2010a,b; Stuiver et al., 2014; Neves et al., 2017; Zuidersma et al., 2020).
Table 2. Impact of Ni2+ exposure on the mineral content of Brassica rapa. 10-day old seedlings were grown on 25% Hoagland nutrient solutions containing supplemental NiCl2 levels of 0, 1, 2 or 5 μM for 10 days. Data on mineral content (μmol g–1 DW) represent the mean of three measurements with three plants in each (± SD). Different letters indicate significant differences between treatments (P ≤ 0.05, one-way ANOVA; Tukey’s HSD test as a post-hoc test).
| NiCl2 concentration (μM) | |||
Mineral content | 0 | 1 | 2 | 5 |
Shoot |
|
|
|
|
Calcium | 766 ± 14a | 761 ± 51a | 781 ± 15a | 815 ± 15a |
Copper | 0.08 ± 0.01a | 0.06 ± 0.01a | 0.06 ± 0.01a | 0.14 ± 0.02b |
Iron | 1.43 ± 0.32a | 1.33 ± 0.06a | 1.01 ± 0.14a | 1.28 ± 0.20a |
Magnesium | 184 ± 2a | 191 ± 10a | 201 ± 1a | 252 ± 8b |
Manganese | 3.1 ± 0.2a | 3.2 ± 0.3a | 2.8 ± 0.1a | 1.9 ± 0.1b |
Nickel | 0.03 ± 0.03a | 0.53 ± 0.03b | 1.04 ± 0.03c | 2.12 ± 0.02d |
Phosphorus | 235 ± 10a | 225 ± 18a | 210 ± 5a | 213 ± 4a |
Sulfur | 249 ± 14a | 257 ± 10a | 258 ± 12a | 241 ± 5a |
Potassium | 1720 ± 42a | 1350 ± 52b | 1375 ± 20b | 1089 ± 22c |
Zinc | 0.54 ± 0.05a | 0.59 ± 0.02a | 0.55 ± 0.01a | 0.70 ± 0.12a |
Root |
|
|
|
|
Calcium | 182 ± 22a | 182 ± 15a | 170 ± 3a | 189 ± 5a |
Copper | 0.29 ± 0.02a | 0.23 ± 0.01b | 0.28 ± 0.02ab | 0.73 ± 0.03c |
Iron | 23.7 ± 0.8a | 34.3 ± 1.9b | 34.9 ± 1.1b | 45.5 ± 3.7c |
Magnesium | 191 ± 12a | 198 ± 9a | 207 ± 22a | 237 ± 26a |
Manganese | 44 ± 3a | 52 ± 2ab | 54 ± 3b | 50 ± 4ab |
Nickel | 0.05 ± 0.01a | 10.9 ± 0.4b | 22.5 ± 1.3c | 33.7 ± 1.9d |
Phosphorus | 322 ± 9a | 321 ± 6a | 316 ± 11a | 305 ± 10a |
Sulfur | 358 ± 3a | 295 ± 7b | 307 ± 9bc | 274 ± 16c |
Potassium | 1556 ± 11a | 1595 ± 67a | 1615 ± 86a | 1319 ± 59b |
Zinc | 0.86 ± 0.16a | 0.74 ± 0.03a | 1.01 ± 0.07a | 2.17 ± 0.40b |
Exposure to elevated Ni2+ concentrations only slightly affected the S status of B. rapa. There was a 20% decrease in the total S content of the root at ≥ 1 μM Ni2+ which could be fully attributed to a decrease in sulfate content (Table 3). Exposure to Ni2+ hardly affected the total sulfur and sulfate content of the shoot (Table 3). In Brassica the uptake of sulfate by the roots is regulated by the expression and activity of the Group 1 sulfate transporters, by viz. Sultr1;2 at sulfate-sufficient and Sultr1;1 and Sultr1;2 at sulfate-deprived conditions (Koralewska et al., 2007). Exposure of Brassica to excessive Zn2+ and Cu2+ resulted in an increased activity of the sulfate transporters and substantially enhanced sulfate content of the shoot (Shahbaz et al., 2010a; Stuiver et al., 2014). Evidently exposure to these toxic metals resulted in a deregulation of the uptake and metabolism of sulfur (Shahbaz et al., 2010a; Stuiver et al., 2014). By contrast, exposure to excessive Mn2+ and MoO42- hardly affected tissue sulfate contents and the sulfate uptake capacity (expressed on a whole-plant fresh weight basis; Neves et al., 2017; Zuidersma et al., 2020). Thus, different heavy metals interfere to different extents with the signal cascade that regulates the sulfate transporters (Shahbaz et al., 2010a; Stuiver et al., 2014; Neves et al., 2017; Zuidersma et al., 2020). Interestingly, the concentration of hydrogen sulfide (H2S) inside cells may regulate the transcription of sulfate transporters (De Kok et al., 2011). Heavy metals may react with H2S, which may reduce cellular H2S levels (Rumble, 2009). However, metals differ in their H2S-reactivity (Rumble, 2009), which may potentially underlie variation in metal impact on the regulation of the sulfate transporters.
Table 3. Impact of Ni2+ exposure on sulfate, water-soluble non-protein and total sulfur content of Brassica rapa. 10-day old seedlings were grown on 25% Hoagland nutrient solutions containing supplemental NiCl2 levels of 0, 1, 2 or 5 μM for 10 days. Data on sulfate and water-soluble non-protein thiol content (μmol g–1 FW) represent the mean of two independent experiments with four to six (sulfate) or five to six (thiols) measurements with three plants in each (± SD). Data on total sulfur content (μmol g–1 FW) were calculated from the data in Table 2 by multiplying for each treatment the contents per g DW with the average dry matter content. Total sulfur data represent the mean of three measurements with three plants in each (± SD). Different letters indicate significant differences between treatments (P ≤ 0.05, one-way ANOVA; Tukey’s HSD test as a post-hoc test).
|
| NiCl2 concentration (μM) | ||
| 0 | 1 | 2 | 5 |
Shoot |
|
|
|
|
Sulfate | 11.2 ± 2.2a | 11.4 ± 1.3a | 11.5 ± 1.0a | 12.1 ± 1.2a |
Thiols | 0.33 ± 0.03a | 0.36 ± 0.10a | 0.37 ± 0.04a | 0.54 ± 0.08b |
Total sulfur | 21.3 ± 1.2a | 20.8 ± 0.8a | 22.9 ± 1.1ab | 25.1 ± 0.5b |
Root |
|
|
|
|
Sulfate | 12.5 ± 1.0a | 8.9 ± 2.5b | 8.6 ± 1.1b | 7.0 ± 1.3b |
Thiols | 0.27 ± 0.07a | 0.29 ± 0.04a | 0.33 ± 0.04a | 0.61 ± 0.11b |
Total sulfur | 21.3 ± 0.2a | 17.2 ± 0.4b | 17.9 ± 0.5b | 17.7 ± 1.0b |
The S-containing compounds cysteine, glutathione and phytochelatins may be important for plant Ni2+ tolerance due to their molecular characteristics (Sirko & Gotor, 2007; Cuypers et al., 2009). However, although exposure to 2 μM Ni2+ strongly inhibited growth of B. rapa, it did not affect the size of the water-soluble non-protein thiol pool (Table 3). Only at 5 μM Ni2+, where the biomass production completely halted, there was a 2.3 and 1.6-fold increased water-soluble non-protein thiol content of the root and shoot, respectively (Table 3). Despite the increase in the content of water-soluble non-protein thiol compounds in the root of B. rapa at 5 μM Ni2+, the absence of any impact of 2 μM Ni2+ on the thiol pool suggests that cysteine, glutathione and phytochelatins are not directly important for Ni2+ tolerance of B. rapa. Notably, this may seem in contrast with earlier experiments, showing that transgenic Arabidopsis thaliana overproducing glutathione had a higher growth rate upon exposure to 100 μM Ni2+ than wildtype plants (Freeman et al., 2004). However, genetic manipulation of the glutathione synthesis pathway may not only alter tissue glutathione content, but also the contents of other metabolites, which may affect Ni2+ tolerance. Accordingly, exposure of Brassica to excessive Mn2+ and MoO42- similarly did not affect the tissue levels of water-soluble non-protein thiols (Neves et al., 2017; Zuidersma et al., 2020). Furthermore, although exposure of Brassica to excessive Zn2+ and Cu2+ enhanced the size of this pool, it was unlikely that this increase had any significance for heavy metal detoxification: experimental manipulation of the size and composition of the thiol pool did not alter the Cu2+ tolerance of Chinese cabbage (Shahbaz et al., 2010a; 2014). Thus, in conclusion, sulfur metabolism is unlikely to have direct relevance for Ni2+ tolerance mechanisms.
T. Ausma was sponsored by a grant (ALW Graduate Program Grant 2017.015) from the Netherlands Organization for Scientific Research and M.J. Hawkesford was funded by the Designing Future Wheat (DFW) project (BB/P016855/1) from the Biotechnology and Biological Sciences Research Council (BBSRC) of the U.K. The authors thank Dr. Mohammad Nawaz and Mrs. Saroj Parmar for their contributions to this research.
The authors declare no conflicts of interest.
Brown, P.H., 2006: Nickel. In: Handbook of Plant Nutrition, 2nd ed., Barker A.V., D.J. Pilbeam (eds.), CRC Press, Boca Raton, Florida, USA, 395-409.
Cataldo, D.A., K.M. McFadden, T.R. Garland, R.E. Wildung, 1988: Organic constituents and complexation of nickel (II), iron (III), cadmium (II) and plutonium (IV) in soybean xylem exudates. Plant Physiology 86, 734-739, DOI: 10.1104/pp.86.3.734.
Cuypers, A., K. Smeets, J. Van Gronsveld, 2009: Heavy metal stress in plants. In: Plant Stress Biology: From Genomics to Systems Biology. Hirt, H. (ed.), Wiley, Weinheim, Germany, 161-178.
De Kok, L.J., F. Buwalda, W. Bosma, 1988: Determination of cysteine and its accumulation in spinach leaf tissue upon exposure to excess sulfur. Journal of Plant Physiology 133, 502-505, DOI: 10.1016/S0176-1617(88)80045-2.
De Kok, L.J., I. Stulen, M.J. Hawkesford, 2011: Sulfur nutrition in crop plants. In: The Molecular Basis of Nutrient Use Efficiency in Crops. Hawkesford, M.J., P. Barraclough, (eds.), Wiley, Oxford, United Kingdom, 295-309.
Fabiano, C.C., T. Tezotto, J.L. Favarin, J.C. Polacco, P. Mazzafera, 2015: Essentiality of nickel in plants: a role in plant stresses. Frontiers in Plant Science, 6, 754, DOI: 10.3389/fpls.2015.00754.
Freeman, J.L., M.W. Persans, K. Nieman, C. Albrecht, W. Peer, I.J. Pickering, D.E. Salt, 2004: Increased glutathione biosynthesis plays a role in nickel tolerance in Thalspi nickel hyperaccumulators. Plant Cell, 16, 2176-2191, DOI: 10.1105/tpc.104.023036.
Gerendas, J., J.C. Polacco, S.K. Freyermuth, B. Sattelmacher, 1999: Significance of nickel for plant growth and metabolism. Journal of Plant Nutrition and Soil Science, 162, 241-256, DOI: 10.1002/(SICI)1522-2624(199906)162:3<241::AID-JPLN241>3.0.CO,2-Q.
Holland, M.A., J.C. Polacco, 1992: Urease-null and hydrogenase-null phenotypes of a phylloplane bacterium reveal altered nickel metabolism in two soybean mutants. Plant Physiology, 98, 942-948, DOI: 10.1104/pp.98.3.942.
Koralewska, A., F.S. Posthumus, C.E.E. Stuiver, P. Buchner, M.J. Hawkesford, L.J. De Kok, 2007: The characteristic high sulphate content in Brassica oleracea is controlled by the expression and activity of sulphate transporters. Plant Biology, 9, 654-661, DOI: 10.1055/s-2007-965438.
Lichtenthaler, H.K., 1987: Chlorophylls and carotenoids: pigments of the photosynthetic biomembranes. Methods in Enzymology, 148, 350-382, DOI: 10.1016/0076-6879(87)48036-1.
Maas, F.M., I. Hoffmann, M.J. Van Harmelen, L.J. De Kok, 1986: Refractometric determination of sulfate and anions in plants separated by high performance liquid chromatography. Plant and Soil, 91, 129-132.
Neves, M.I., D.H. Prajapati, S. Parmar, T. Aghajanzadeh, M.J. Hawkesford, L.J. De Kok, 2017: Manganese toxicity hardly affects sulfur metabolism in Brassica rapa. In: Sulfur Metabolism in Higher Plants – Fundamental, Environmental and Agricultural Aspects. De Kok, L.J., M.J. Hawkesford, S.H. Haneklaus, E. Schnug (eds.), Springer, Dordrecht, The Netherlands, 155-162.
Pilon, M., C.M. Cohu, K. Ravet, S.E. Abdel-Ghany, F. Gaymard, 2009: Essential transition metal homeostasis in plants. Current Opinion in Plant Biology, 12, 347-357, DOI: 10.1016/j.pbi.2009.04.011.
Polacco, J.C., 1977: Nitrogen metabolism in soybean tissue culture: II. Urea utilization and urease synthesis requires Ni2+. Plant Physiology, 59, 827-830, DOI: 10.1104/pp.59.5.827.
Polacco, J.C., P. Mazzafera, T. Tezotto, 2013: Opinion – Nickel and urease in plants: still many knowledge gaps. Plant Science, 199, 79-90, DOI: 10.1016/j.plantsci.2012.10.010.
Reich, M., T. Aghajanzadeh, J. Helm, S. Parmar, M.J. Hawkesford, L.J. De Kok, 2017: Chloride and sulfate salinity differently affect biomass, mineral nutrient composition and expression of sulfate transport and assimilation genes in Brassica rapa. Plant and Soil, 411, 319-332, DOI: 10.1007/s11104-016-3026-7.
Reis, A.R., J.P.Q. Barcelos, C.R.W.S. Osório, E.F. Santos, L.A.M. Lisboa, J.M.K. Santini, M. Jose, D. Santos, E.F. Junior, M. Campos, P.A.M. Figueiredo, J. Lavres, P.L. Gratao, 2017: A glimpse into the physiological, biochemical and nutritional status of soybean plants under Ni-stress conditions. Environment and Experimental Botany, 144, 76-87, DOI: 10.1016/j.envexpbot.2017.10.006.
Rumble, J.R., 2009: CRC Handbook of chemistry and physics, 90th ed., CRC Press, Boca Raton, Florida, United States.
Shahbaz, M., M.H. Tseng, C.E.E. Stuiver, A. Koralewska, F.S. Posthumus, J.H. Venema, S. Parmar, H. Schat, M.J. Hawkesford, L.J. de Kok, 2010a: Copper exposure interferes with the regulation of the uptake, distribution and metabolism of sulfate in Chinese cabbage. J. Plant Physiology, 167, 438-446, DOI: 10.1016/j.jplph.2009.10.016.
Shahbaz, M., M.H. Tseng, C.E.E. Stuiver, F.S. Posthumus, S. Parmar, A. Koralewska, M.J. Hawkesford, L.J. De Kok, 2010b: Impact of copper exposure on physiological functioning of Chinese cabbage. In More Sustainability in Agriculture: New Fertilizers and Fertilizer Management. Sequi, P., D. Ferri, E. Rea, A.V. Montemurro, F. Fornado (eds.), Fertilitas Agronum, Rome, Italy, 318-324.
Shahbaz, M., S. Parmar, C.E.E. Stuiver, M.J. Hawkesford, L.J. De Kok, 2014: Copper toxicity in Chinese cabbage is not influenced by plant sulphur status, but affects sulphur metabolism-related gene expression and the suggested regulatory metabolites. Plant Biology, 16, 68-78, DOI: 10.1111/plb.12019.
Shahzad, B., M. Tanveer, A. Rehman, S. Alam Cheema, S. Fahad, S. Rehman, A. Sharma, 2018: Nickel, whether toxic or essential for plants and environment – a review. Plant Physiology and Biochemistry, 132, 641-651, DOI: 10.1016/j.plaphy.2018.10.014.
Sirko, A., C. Gotor, 2007: Molecular links between metals in the environment and plant sulfur metabolism. In: Sulfur in Plants – an Ecological Perspective. Hawkesford, M.J., L.J. De Kok (eds.), Springer, Dordrecht, The Netherlands, 169-195.
Stuiver, C.E.E., F.S. Posthumus, S. Parmar, M. Shahbaz, M.J. Hawkesford, L.J. De Kok, 2014: Zinc exposure has differential effects on uptake and metabolism of sulfur and nitrogen in Chinese cabbage. Journal of Plant Nutrition and Soil Science, 177, 748-757, DOI: 10.1002/jpln.201300369.
Wood, B.W., C.C. Reilly, A.P. Nyczepir, 2004: Mouse-ear of Pecan: A nickel deficiency. Horticultural Science, 39, 1238-1242, DOI: 10.21273/HORTSCI.39.6.1238.
Yadav, S.K., 2010: Heavy metals toxicity in plants: an overview on the role of glutathione and phytochelatins in heavy metal stress tolerance in plants. South African Journal of Botany, 76, 167-179, DOI: 10.1016/j.sajb.2009.10.007.
Zuidersma, E.I., T. Ausma, C.E.E. Stuiver, D.H. Prajapati, M.J. Hawkesford, L.J. De Kok, 2020: Molybdate toxicity in Chinese cabbage is not the direct consequence of changes in sulphur metabolism. Plant Biology, 22, 331-336, DOI: 10.1111/plb.13065.