

Agronomic evaluation of bone char as phosphorus fertiliser after five years of consecutive application
Agronomische Bewertung nach fünfjähriger Phosphordüngung mit Knochenkohle
Journal für Kulturpflanzen, 72 (12). S. 561–576, 2020, ISSN 1867-0911, DOI: 10.5073/JfK.2020.12.02, Verlag Eugen Ulmer KG, Stuttgart
 | This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/deed.en). Dies ist ein Open-Access-Artikel, der unter den Bedingungen der Creative Commons Namensnennung 4.0 International Lizenz (CC BY 4.0) zur Verfügung gestellt wird (https://creativecommons.org/licenses/by/4.0/deed.de). |
Knochenkohle (BC), hergestellt mittels einer technischen Pyrolyse von entfetteten Tierknochen aus Schlachtabfällen, könnte ein vielversprechender Phosphor (P) Recyclingdünger sein. Agronomische Bewertungen aus Langzeitversuchen fehlen jedoch bisher. Phosphor in BC (15% P) ist überwiegend in Strukturen ähnlich dem Hydroxylapatit (HA) gebunden. Es kann davon ausgegangen werden, dass in HA gebundener P aufgrund geringer Löslichkeit keine große Düngewirkung erzielt. Eine bessere Löslichkeit wird von Schwefel angereicherter BC (BCplus) erwartet.
2013 wurde ein Feldversuch angelegt, um das Düngepotenzial von BC und BCplus im Vergleich zu P0 und Triple Superphosphat (TSP) zu testen. In Abhängigkeit der durchschnittlichen PCAL-Konzentration im Boden (0–30 cm) wurden Parzellen den Boden-P-Gehaltsklasse A (11 mg kg–1 PCAL), B (21 mg kg–1 PCAL), und C (47 mg kg–1 PCAL) zugeordnet. In Boden-P-Gehaltklasse A stieg der mittlere Relativertrag kumuliert für die erste Fruchtfolge (Wintergerste, Winterraps, Winterweizen, Lupine, Winterroggen) in der Reihenfolge P0 (90 %) < BC (94 %) < BCplus (95 %) < TSP (100 %) an. Die relative P-Aufnahme (Korn) wurde durch die Düngevarianten in Boden-P-Gehaltsklasse A in der Reihenfolge P0 (81 %) < BC (88 %) < BCplus (91 %) < TSP (100 %) und in Klasse B von P0 (84 %) < BCplus (90 %) < BC (92 %) < TSP (100 %) beeinflusst. Bei ausreichender P-Versorgung (Boden-P-Gehaltsklasse C) wurde im Fruchtfolgemittel kein Düngereinfluss auf die Ertragsbildung und die P-Aufnahme beobachtet. Die PCAL-Konzentration im Boden wurde ausschließlich durch die Anwendung von TSP erhöht. Ausgehend von einer ausreichenden P-Versorgung (Boden-P-Gehaltsklasse C) werden weitere Versuchsjahre zeigen, ob Knochenkohlen in der Lage sind, die Nährstoffversorgung von Kulturpflanzen mit P langfristig zu gewährleisten.
Stichwörter: Knochenkohle, Phosphorrecycling, Recyclingdünger, Pflanzenernährung
Bone char (BC), pyrolyzed defatted bones from slaughterhouse waste, may be a promising secondary raw material to produce phosphorus (P) fertiliser, but its agronomic value has not yet been shown in longer-term field experiments. Phosphorus in BC (15 % P) is mainly bound in a structure similar to hydroxylapatite (HA). Generally, P in HA is of low solubility and, consequently, it can be expected that bones or BC have a low fertilisation value. Better solubility of P is expected from BC enriched with sulphur (BCplus). In 2013, a field experiment was established to test the fertilisation potential of BC and BCplus compared to a control (P0) and triple super phosphate (TSP) treatment. According to their mean PCAL concentrations (0–30 cm) plots were assigned to the initial soil P-test class (iSPTC) A (11 mg kg–1 PCAL), B (21 mg kg–1 PCAL), and C (47 mg kg–1 PCAL). After a first crop rotation an increased cumulative yield was determined for iSPTC-A depending on the fertiliser treatment with means of relative yield of P0 (90 %) < BC (94 %) < BCplus (95 %) < TSP (100 %). Effects on the relative P uptake of grain in iSPTC-A depending on the fertiliser treatment were in the order of P0 (81 %) < BC (88 %) < BCplus (91 %) < TSP (100 %) and in iSPTC-B of P0 (84 %) < BCplus (90 %) < BC (92 %) < TSP (100 %). Fertiliser treatments had no effect on mean yield and P uptake in the first crop rotation if initially sufficient soil P (iSPTC-C) was available. An increase of PCAL concentrations was only achieved by TSP fertilisation. Future experimental years will show, if BC fertilisers are able to maintain sufficient P availability to crops in the long term.
Key words: Bone char, phosphorus recycling, recycling fertiliser, plant nutrition
Phosphate rock reserves are limited and there are predictions that the global peak of phosphorus production will be around 2030 (Cordell et al., 2009). Estimations about the total depletion of phosphate reserves are difficult and uncertain because of continuously changing boundary conditions such as, the world population with changing dietary patterns, losses in fertiliser production and food chains, and changing assessments of phosphate reserves (Neset & Cordell, 2012). Even though a shortage of P in the next decades is unlikely (Scholz & Wellmer, 2013), losses from the anthropogenic use chain should be minimized to extend/maintain the availability of phosphate resources for future generations and protect environmental compartments such as aquatic and marine ecosystems. van Dijk et al. (2016) estimated that recovered P in the European Union (EU-27) could replace 43 % of the actually applied mineral P fertiliser. On the other hand, about half (924 Gg) of the imported P accumulates in agricultural soils of the EU-27 annually (Buckwell & Nadeu, 2016). This increases the risk of environmental burdens mainly through P transport by erosion and leaching processes to surface waters and subsequent eutrophication (Withers & Haygarth, 2007; Kleinman et al., 2011; Ulén et al., 2012; Smith et al., 2019).
Phosphorus is an essential plant macronutrient and cannot be replaced by any other chemical element; hence, agriculture has a high demand of P fertilisers. Therefore, most (82 %) of the mined phosphate rock is transformed into P fertilisers (Scholz & Wellmer, 2013). Since phosphate rock was inserted into a European list of critical raw materials in 2014 (EU, 2014), efforts have increased substantially to replace rock phosphate by recycled P from, e.g., sewage sludge, waste streams, and animal by-products.
Animal bones as P resource are exceptional in that they contain high amounts of P as well as calcium (Ca) and magnesium (Mg), both major plant nutrient elements, but have very low concentrations of heavy metals (Cascarosa et al., 2012). After the bovine spongiform encephalopathy (BSE) crisis had resulted in bans of meat and bone meal use in animal feeding in the EU in 1994, technically pyrolyzed bone material, i.e. bone char (BC), has been considered as potential P source in cropping (Vassilev et al., 2013). This included research in potential use as carrier for beneficial microorganisms (Postma et al., 2013) and phosphate fertiliser (Warren et al., 2009), the latter avoiding or even immobilising harmful metals such as Cd in soil (Siebers et al., 2014). While a P mobilisation potential has been demonstrated in various laboratory incubation experiments (Morshedizad et al., 2016; Zwetsloot et al., 2016), pot experiments with bone char sometimes revealed low P-uptake by crops (Siebers et al., 2012; Siebers et al., 2014). Warren et al. (2009) showed that the P release from BC strongly depends on soil pH. In that publication, soil with pH ranging from 3.4 to 8.8 were incubated with BC for 145 days and whilst acidic soils dissolved nearly 70 % P, neutral to alkaline soils did not dissolve any P, confirming that soil acidity affects P release from BC (Warren et al., 2009). Therefore, encrusting of BC particles with reduced S-compounds has been tested as an approach to improve the phosphate mobilisation and potential agronomic efficiency by oxidation and subsequent acid production in soil to enhance the P-release. In this research Zimmer et al. (2018) demonstrated spectroscopically and spectro-microscopically that elemental and P-/S-species distributions in BCplus particles (“plus” for the S-addition) or on their surface may favour the “in situ digestion” (Fan et al., 2002) of the source BC by oxidation of reduced S compounds, including elemental S. However, long-term P fertilisation effects of neither BCs nor other P-recycling fertilisers have been studied in field experiments. The agronomic efficiency of new P fertilisers from waste streams is largely unknown, and especially experience from multi-annual field studies is lacking.
To overcome this knowledge gap, a field experiment was carried out to answer the following questions: (a) Do BCs have any agronomic value for supplying P to crops and how do they do in comparison with traditional mineral P fertiliser, (b) To what extent do the additional S components of BCplus increase the plant availability of P and, thus, increase the P-uptake by plants and crop yield under field conditions, and (c) Does the initial P status of experimental soil affect the P solubility of original BC and BCplus?
The field experiment was located in Braunschweig, Lower Saxony, Germany (52°18'N 10°27'E), 81 m above sea level; the site was characterized by a mean annual precipitation of 620 mm and a mean annual temperature of 9°C. Soils at the experimental field were classified as Dystric Cambisol and Haplic Luvisol (IUSS Working Group WRB, 2014) and were built from sandy loess overlying sandy fluviatile sediments. Top soil (0–30 cm) had an average total carbon content of 1.37 % and consisted of 36 % sand, 57 % silt, and 7 % clay at the beginning of the experiment in 2013. The trial was set up in an experimental site where a former long-term P field experiment from 1985 to 2008 (Vogeler et al., 2009) left differences in concentrations of plant available soil P. Briefly, from 1985–1997 mineral P fertiliser was annually applied (n = 4) in treatment (T) T1 = 0P, T2 = 21.8 kg P ha–1 in spring, T3 = 21.8 kg P ha–1 in fall and spring each, T4 = P uptake by previous crop, T5 = P uptake by previous crop × 1.5; from 1998–2008 (n = 3, half of the plots under conventional tillage, half under conservation tillage) T1 = 0P, T2 = P applied through farm yard manure (FYM) only, T3 = 45 kg P ha–1 mineral P and FYM P; from 2009–2012 (n = 3, grassland) T1 = 0P, T2 = 0P, T3 = 30 kg mineral P ha–1. In spring 2013, the experiment was ploughed to a depth of 25 cm and oat was sown. According to the contents of plant available P (PCAL) after harvest 2013 plots were assigned to so-called iSPTCs (initial soil P-test classes) A (severely deficient, < 15 mg PCAL kg–1), B (deficient, 15–30 mg PCAL kg–1), and C (sufficient, 31–60 mg PCAL kg–1), respectively (Wiesler et al., 2018). Table 1 provides an overview about initial soil P, N and C contents as well as pH of the plots before the start of the field experiment.
Table 1. Means of basic soil parameters (0–30 cm) across plots with similar initial soil P-test class (iSPTC) in 2013 (pre-experiment)
Soil parameter | iSPTC-A | iSPTC-B | iSPTC-C |
pH | 5.1 | 5.2 | 5.2 |
Pwater [mg kg–1] | 2.8 | 4.4 | 9.4 |
PCAL [mg kg–1] | 11.2 | 20.7 | 47.2 |
Total P [mg kg–1] | 217 | 273 | 399 |
Total C [%] | 1.3 | 1.4 | 1.4 |
Total N [%] | 0.10 | 0.10 | 0.10 |
In autumn 2013, the experiment was established in a completely randomized block design with iSPTCs been already randomly available from the former experiment. Fertilizer types were assigned randomly in each iSPTC maintaining the original three blocks. The plots had a size of 5.75 m × 17.5 m (original plots were split in half), and a crop rotation of winter barley, winter oilseed rape, winter wheat, lupine and winter rye (Table 2). All straw remained on the field and was therefore not taken into account for P budgeting and uptake. Annually, chisel ploughing and ploughing to a depth of 25 cm incorporated straw and crop residues before seeding. Table 2 provides an overview of agronomic measures in the period 2013 to 2018.
Table 2. Crop/varieties grown and timing of agronomic measures during the first crop rotation of the experiment from 2013 to 2018
Harvest year | 2014 | 2015 | 2016 | 2017 | 2018 |
Crop | Winter barley | Winter oilseed rape | Winter wheat | Lupine | Winter rye |
Binomial name | Hordeum vulgare L. | Brassica napus L. | Triticum aestivum L. | Lupinus angustifolius L. | Secale cereale L. |
Variety | Otto | Arsenal | JB Asano | Boregine | Daniello |
Chisel ploughing dates | 03.09.13 | 24.07.+08.08.14 | 12.08.15 | 20.09.+18.10.16 | 13.09.17 |
Ploughing date | 20.09.13 | 22.08.14 | 05.10.15 | 23.03.17 | 17.10.17 |
P application date | 23.09.13 | 25.08.14 | 12.10.15 | 27.03.17 | 18.10.17 |
Seeding date | 26.09.13 | 01.09.14 | 13.10.15 | 28.03.17 | 19.10.17 |
Total N appl. [kg ha–1] | 160 | 160 | 130 | – | 120 |
Total K appl. [kg ha–1] | 100 | 150 | 100 | 133 | 100 |
Total S appl. [kg ha–1]* | – | 40 | 12 | 20 | 15 |
Total Mg appl. [kg ha–1]* | 41 | – | 11 | 79 | 21 |
Total Ca appl. [kg ha–1]* | 909 | – | – | 1441 |
|
Spring sampling date/BBCH# | – | – | 12.04.16/BBCH 32 | 12.06.17/BBCH 65 | 27.04.18/BBCH 32 |
Harvest date | 15.07.14 | 23.07.15 | 28.07.16 | 04.09.17 | 19.07.18 |
* Total S, Mg, and Ca application without S, Mg, and Ca components of P-fertilisers. | |||||
Three different types of P fertiliser were applied to an equivalent of 45 kg P ha–1 once a year shortly before seeding and a control without P for comparison: bone char (BC) pyrolysed at more than 800°C, surface-modified bone char (BCplus) with sulphur compounds from biogas streams (patent DE102011010525), and highly water soluble triple super phosphate (TSP). Fertilisers were used as provided with 95–100 % of the particles bigger than 1 mm. Total concentration of the fertilizer elements were extracted by Aqua regia (VDLUFA, 2000) and P (177.4 nm), Ca (318.1 nm), K (766.4 nm), Mg (279.0 nm), and Na (589.5 nm) were measured by Inductively Coupled Plasma-Optical Emission Spectrometry (ICP-OES, icap 6000, Thermo Fisher, Cambridge, United Kingdom), As, Cd, Cr, Cu, Ni, Pb, U, and Zn by High-Resolution Inductively Coupled Plasma Mass Spectrometry (ICP-MS, Thermo Element XR, Thermo Fisher, Cambridge, United Kingdom). BC and BCplus had much lower concentrations of As, Cd, Cr, Cu, Ni, and U than TSP fertiliser (Table 3). Further detailed information about the three fertilizers was provided by Zimmer et al. (2019).
Table 3. Total element concentrations in Aqua regia extracts (VDLUFA, 2000) of the three tested fertilizers BC, BCplus and TSP
Element | BC | BCplus | TSP |
P [%] | 14.81 | 10.72 | 20.04 |
Ca [%] | 32.08 | 23.54 | 15.86 |
K [%] | 0.10 | 0.03 | 0.08 |
Mg [%] | 0.60 | 0.40 | 0.65 |
Na [%] | 0.80 | 0.51 | 0.28 |
As [mg kg–1] | 0.10 | 0.25 | 4.25 |
Cd [mg kg–1] | < 0.08 | < 0.02 | 12.50 |
Cr [mg kg–1] | 1.30 | 0.27 | 203.23 |
Cu [mg kg–1] | 1.41 | 4.42 | 18.29 |
Ni [mg kg–1] | 0.41 | 0.28 | 25.34 |
Pb [mg kg–1] | 2.21 | 0.70 | 1.77 |
U [mg kg–1] | 0.09 | < 0.02 | 112.22 |
Zn [mg kg–1] | 116.76 | 69.90 | 376.89 |
Each year (2014–2018) after crop harvest, eight soil cores per plot were taken to a depth of 30 cm and combined to a bulk soil sample. These samples were air-dried and sieved < 2 mm. During sampling in spring 2016–2018 (Table 2), 10 soil cores per plot were taken to a depth of 10 cm and combined. Half of that soil was immediately cooled and stored at 4°C until further analyses of alkaline and acidic phosphatases. The other half of that soil sample was air dried and sieved < 2 mm.
From dried soil samples, soil pH was determined in 0.01 M CaCl2 (10 g soil in 25 ml CaCl2) and water soluble P (Pwater) was extracted in a procedure slightly modified from van der Paauw et al. (1971) (1.5 g soil in 2 ml aqua dest. for 22 hours, addition of 70 ml aqua dest., overhead shaking for 60 min) before P was analysed colorimetrically (Specord 50, Analytik Jena, Germany) using the molybdenum blue method at a wavelength of 882 nm according to Murphy & Riley (1962). Plant available P (PCAL) was extracted from soil with calcium acetate lactate (Schüller, 1969) and total soil P concentration (Ptot) was extracted by Aqua regia (VDLUFA, 1991). In both extracts, P was measured at a wavelength of 213.6 nm using Inductively Coupled Plasma-Optical Emission Spectrometry (ICP-OES; icap 6000, Thermo Fisher, Cambridge, United Kingdom).
From the spring samples (0–10 cm) potential acidic and alkaline phosphatase activity was determined according to Schinner et al. (1996) based on the method developed by Tabatabai & Bremner (1969) and Eivazi & Tabatabai (1977). The enzyme activities of 1 g field moist soil after 1 h of incubation at 37°C were measured as p-nitrophenol coloured by 0.5 m L–1 sodium hydroxide; colorimetric analysis was carried out at 400 nm (Specord 50, Analytik Jena, Germany).
At harvest (2014–2018), central areas of the plots were harvested by a combine (Hege 160, cutting width 1.5 m × 16 m length) and fresh grain yield was recorded by the balance of the combine. Subsamples of the grains were taken. During the sampling campaigns in spring (2016–2018), four times 0.25 m2 per plot were cut and combined to measure the fresh matter (FM) yield per plot. Plant samples were dried at 60°C until constant weight in a ventilated oven to determine dry matter (DM) yield per plot. Dried samples were ground ≤ 0.5 mm with an ultracentrifugal mill (Retsch ZM 100 or 200, Haan, Germany). Plant material (0.5 g) was digested with 6 ml nitric acid and 1.5 ml hydrogen peroxide in a microwave (CEM MARS, Metthews, USA) to determine the P concentration with ICP-OES (icap 6000, Thermo Fisher, Cambridge, United Kingdom) at a wavelength of 177.4 nm.
Statistical analyses were performed with R version 3.5.1 (R Core Team, 2018). The effects of three iSPTCs, four fertiliser treatments and their interaction on yield, P-uptake by plants and soil parameters were analysed by a two-factor analysis of variance (ANOVA) with the ‘aov’-function. The combination of the three iSPTCs and four fertiliser treatments on P uptake was also analysed by a one-factor ANOVA with the ‘aov’-function and a post-hoc test with the ‘TukeyHSD’-function.
Mean relative grain yield and mean relative P uptake of grain in relation to TSP fertilisation (100 %) from 2014–2018 was calculated according to Eq. 1 and 2.
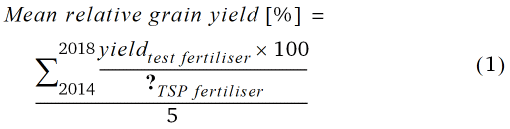
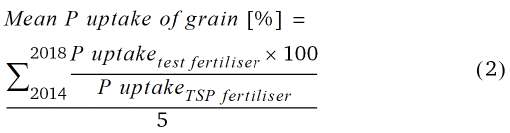
For the first crop rotation (2014–2018), the apparent nutrient recovery efficiency of grain (ANR; (Baligar et al., 2001)) and the P budget were calculated according to Eq. 3 and 4, respectively.
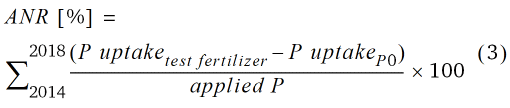
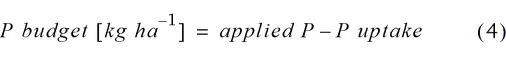
In three experimental years (2016–2018), significant differences in P uptake were recorded in spring (Table 4). The P uptake was affected by fertiliser type and iSPTC. Especially at low soil P concentrations (iSPTC-A and -B), the P uptake often followed the order BC < P0 < BCplus < TSP. In none of the years, statistical evidence of any interaction of fertiliser type with iSPTC was determined (Table 4). The P uptake in spring mirrored the Pwater concentration in soils, generally showing the largest concentrations after application of highly water soluble TSP (Table 5). The concentration of Pwater and P uptake correlated for winter wheat (r2 = 0.7961), lupine (r2 = 0.4997) and winter rye (r2 = 0.6797) without significant interaction between fertiliser type and iSPTC (Table 5). The iSPTC or the applied fertiliser type did not affect the acidic and alkaline phosphatase activities (Suppl. 1 and 2). These enzymes had mean potential activities of 68 and 270 μg p-NP g–1 h–1 in 2016, of 73 and 300 μg p-NP g–1 h–1 in 2017, and of 77 and 251 μg p-NP g–1 h–1 in 2018, for the alkaline and the acidic enzyme, respectively (Appendix Table 1 and 2).
Table 4. P uptake in spring 2016 – 2018 as affected by initial soil P-test class (iSPTC; A = severely deficient, B = deficient, C = sufficient) and type of P-fertiliser (P0, BC, BCplus, TSP) (means of three replicates and standard deviation in brackets)
Year | iSPTC | P uptake in spring [kg ha–1] | |||
P0 | BC | BCplus | TSP | ||
2016 | A | 1.7 (0.57) | 1.2 (0.15) | 2.3 (0.61) | 2.6 (0.52) |
B | 2.2 (0.43) | 1.9 (0.37) | 2.8 (0.33) | 3.5 (0.75) | |
C | 3.3 (0.44) | 3.6 (0.31) | 3.6 (0.14) | 4.3 (0.08) | |
2017 | A | 8.9 (0.94) | 7.0 (0.60) | 8.7 (2.74) | 8.9 (1.24) |
B | 10.3 (0.68) | 8.5 (0.57) | 9.9 (1.44) | 11.7 (1.17) | |
C | 11.8 (1.08) | 11.0 (1.45) | 11.1 (1.93) | 11.8 (0.91) | |
2018 | A | 4.9 (2.77) | 3.9 (1.02) | 6.6 (1.58) | 9.1 (2.09) |
B | 6.7 (0.98) | 6.7 (2.07) | 8.7 (1.93) | 8.9 (0.30) | |
C | 9.6 (0.95) | 8.8 (2.35) | 7.8 (0.61) | 10.5 (0.41) | |
| ANOVA | df | F value | P value | Significance# |
2016 | iSPTC (S) | 2 | 44.378 | < 0.001 | *** |
Fertiliser type (F) | 3 | 13.664 | < 0.001 | *** | |
S × F | 6 | 0.878 | 0.527 | n.s. | |
2017 | iSPTC (S) | 2 | 15.104 | < 0.001 | *** |
Fertiliser type (F) | 3 | 3.331 | 0.038 | * | |
S × F | 6 | 0.531 | 0.779 | n.s. | |
2018 | iSPTC (S) | 2 | 10.910 | < 0.001 | *** |
Fertiliser type (F) | 3 | 5.973 | 0.004 | ** | |
S × F | 6 | 1.666 | 0.177 | n.s. | |
#Significance of p-levels: n.s. = not significant (p > 0.05), * = 5 % (p ≤ 0.05), ** = 1 % (p ≤ 0.01), *** = 0.1 % (p ≤ 0.001). | |||||
Table 5. Water-extractable soil P concentration in spring 2016 – 2018 as affected by initial soil P-test class (iSPTC; A = severely deficient, B = deficient, C = sufficient) and type of P-fertiliser (P0, BC, BCplus, TSP) (means of three replicates and standard deviation in brackets)
Year | iSPTC | Pwater in spring [mg kg–1] | |||
P0 | BC | BCplus | TSP | ||
2016 | A | 3.8 (2.25) | 2.3 (0.18) | 3.9 (1.24) | 4.6 (1.30) |
B | 3.9 (0.49) | 3.5 (0.91) | 4.9 (0.47) | 6.2 (1.12) | |
C | 5.7 (1.25) | 6.6 (1.47) | 6.7 (1.35) | 10.4 (1.61) | |
2017 | A | 1.9 (1.07) | 1.2 (0.18) | 2.2 (1.00) | 5.0 (0.32) |
B | 2.1 (0.36) | 2.3 (0.43) | 2.6 (1.10) | 7.2 (3.29) | |
C | 5.6 (0.73) | 6.3 (1.42) | 6.0 (1.48) | 11.8 (0.91) | |
2018 | A | 2.9 (1.36) | 2.2 (0.10) | 3.1 (0.58) | 6.5 (1.02) |
B | 3.8 (0.32) | 3.2 (0.12) | 4.4 (0.13) | 6.6 (0.99) | |
C | 6.5 (0.40) | 7.6 (1.89) | 6.8 (1.20) | 11.1 (2.40) | |
| ANOVA | df | F value | P value | Significance# |
2016 | iSPTC (S) | 2 | 26.984 | < 0.001 | *** |
Fertiliser type (F) | 3 | 9.333 | < 0.001 | *** | |
S × F | 6 | 1.441 | 0.244 | n.s. | |
2017 | iSPTC (S) | 2 | 47.058 | < 0.001 | *** |
Fertiliser type (F) | 3 | 28.078 | < 0.001 | *** | |
S × F | 6 | 0.703 | 0.650 | n.s. | |
2018 | iSPTC (S) | 2 | 53.593 | < 0.001 | *** |
Fertiliser type (F) | 3 | 24.999 | < 0.001 | *** | |
S × F | 6 | 0.912 | 0.505 | n.s. | |
#Significance of p-levels: n.s. = not significant (p > 0.05), * = 5 % (p ≤ 0.05), ** = 1 % (p ≤ 0.01), *** = 0.1 % (p ≤ 0.001). | |||||
Significant effects of the iSPTC on grain yield were recorded in the 1st, 3rd, and 4th year and effects of fertiliser type only in the 1st year (2014) of the experiment (Table 6). Interacting effects of fertiliser type with iSPTC on yield were not observed (Table 6). Summarizing the mean relative yield (Eq. 1) in relation to TSP fertilisation of all years (Fig. 1) showed significant effects of the iSPTC (P value = 0.0021) but there was no statistical evidence of the fertiliser treatments (P value = 0.2718) or the interaction of iSPTC and applied fertiliser type (P value = 0.3616). Only for iSPTC-A a yield effect of fertiliser was obvious and means of relative yield followed the order P0 (90 %) < BC (94 %) < BCplus (95 %) < TSP (100 %) (Fig. 1).
Table 6. Yield response to P fertilisation as affected by initial soil P-test class (iSPTC; A = severely deficient, B = deficient, C = sufficient) and type of P-fertiliser (P0, BC, BCplus, TSP) during the first crop rotation of the experiment from 2014–2018 (means of three replicates and standard deviation in brackets)
Year | iSPTC | Grain yield DM [t ha–1] | |||
P0 | BC | BCplus | TSP | ||
2014 | A | 6.3 (0.61) | 6.1 (0.08) | 6.6 (0.78) | 7.1 (0.20) |
B | 6.7 (0.31) | 6.5 (0.70) | 7.2 (0.44) | 7.3 (0.34) | |
C | 6.8 (0.08) | 6.6 (0.01) | 7.4 (0.13) | 7.2 (0.32) | |
2015 | A | 3.1 (0.86) | 3.8 (0.36) | 3.7 (0.76) | 3.8 (0.45) |
B | 2.9 (0.62) | 3.5 (0.24) | 2.9 (0.78) | 3.7 (0.32) | |
C | 3.2 (0.46) | 3.5 (0.43) | 3.2 (0.28) | 3.0 (0.27) | |
2016 | A | 5.4 (0.25) | 5.4 (0.28) | 5.5 (0.33) | 5.9 (0.12) |
B | 5.7 (0.08) | 5.4 (0.26) | 6.0 (0.16) | 5.7 (0.35) | |
C | 5.7 (0.49) | 5.8 (0.17) | 6.0 (0.29) | 6.0 (0.10) | |
2017 | A | 2.5 (0.26) | 2.7 (0.10) | 2.6 (0.48) | 2.7 (0.24) |
B | 2.5 (0.14) | 2.9 (0.15) | 2.4 (0.27) | 2.4 (0.39) | |
C | 2.4 (0.58) | 2.3 (0.27) | 2.4 (0.31) | 2.1 (0.11) | |
2018 | A | 6.4 (1.08) | 6.3 (0.44) | 6.5 (0.78) | 6.9 (0.34) |
B | 7.1 (0.10) | 6.4 (0.35) | 7.0 (0.39) | 6.6 (0.23) | |
C | 6.8 (0.57) | 7.2 (0.27) | 6.7 (0.64) | 7.2 (0.05) | |
| ANOVA | df | F value | P value | Significance# |
2014 | iSPTC (S) | 2 | 4.788 | 0.019 | * |
Fertiliser type (F) | 3 | 7.699 | 0.001 | ** | |
S × F | 6 | 0.351 | 0.902 | n.s. | |
2015 | iSPTC (S) | 2 | 1.546 | 0.235 | n.s. |
Fertiliser type (F) | 3 | 1.773 | 0.182 | n.s. | |
S × F | 6 | 0.720 | 0.638 | n.s. | |
2016 | iSPTC (S) | 2 | 3.883 | 0.036 | * |
Fertiliser type (F) | 3 | 2.669 | 0.073 | n.s. | |
S × F | 6 | 1.204 | 0.341 | n.s. | |
2017 | iSPTC (S) | 2 | 4.438 | 0.024 | * |
Fertiliser type (F) | 3 | 1.113 | 0.365 | n.s. | |
S × F | 6 | 0.980 | 0.462 | n.s. | |
2018 | iSPTC (S) | 2 | 1.703 | 0.205 | n.s. |
Fertiliser type (F) | 3 | 0.399 | 0.755 | n.s. | |
S × F | 6 | 1.087 | 0.401 | n.s. | |
#Significance of p-levels: n.s. = not significant (p > 0.05), * = 5 % (p ≤ 0.05), ** = 1 % (p ≤ 0.01), *** = 0.1 % (p ≤ 0.001). | |||||
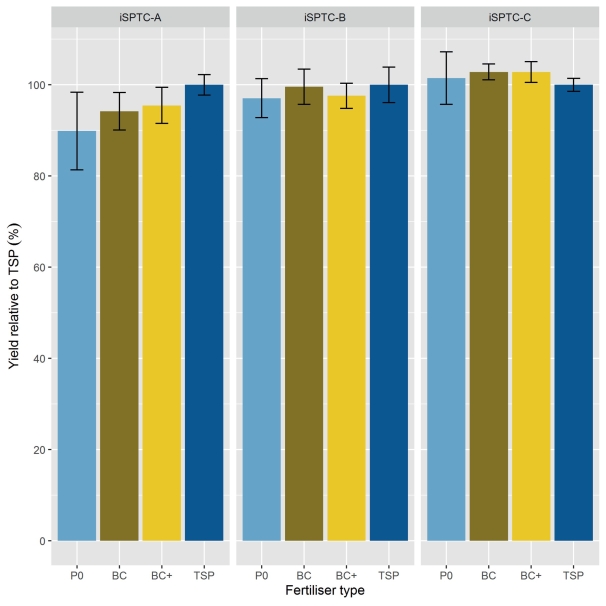
Fig. 1. Mean relative grain yield with TSP = 100 % as affected by initial soil P-test class (iSPTC; A = severely deficient, B = deficient, C = sufficient) and type of P-fertiliser (P0, BC, BC+ (= BCplus), TSP) during the first crop rotation of the experiment from 2014–2018.
P uptake by grain was affected by the iSPTC in the 1st, 3rd, and 5th and by fertiliser type in the 2nd and 5th year of the experiment (Table 7). Even though in the 5th year (2018, winter rye) individual significant effects of iSPTC and fertiliser type on the P uptake by grains were observed, there was no statistical evidence of an interaction of these in any of the years (Table 7). Mean relative P uptake by grain in relation to TSP fertilisation of all years (Fig. 2) showed significant effects of the iSPTC (P value = 0.0017) as well as of the fertiliser treatments (P value = 0.0004) but no statistical evidence of any interaction of iSPTC and applied fertiliser type (P value = 0.2475) were found. Effects were most pronounced in iSPTC-A and -B, with increasing effects on the P uptake by grain in class A depending on the fertiliser treatment: P0 (81 %) < BC (88 %) < BCplus (91 %) < TSP (100 %) and in class B: P0 (84 %) < BCplus (90 %) < BC (92 %) < TSP (100 %) (Fig. 2).
Table 7. P uptake by grains as affected by initial soil P-test class (iSPTC; A = severely deficient, B = deficient, C = sufficient) and type of P-fertiliser (P0, BC, BCplus, TSP) during the first crop rotation of the experiment from 2014–2018 (means of three replicates and standard deviation in brackets)
Year | iSPTC | P uptake by grain [kg ha–1] | |||
P0 | BC | BCplus | TSP | ||
2014 | A | 17.5 (3.33) | 15.6 (0.39) | 17.2 (2.55) | 18.1 (0.47) |
B | 19.8 (0.79) | 18.0 (1.76) | 18.9 (1.21) | 19.8 (1.15) | |
C | 21.1 (1.32) | 21.0 (0.46) | 20.6 (0.74) | 22.7 (0.28) | |
2015 | A | 14.5 (4.65) | 23.5 (0.65) | 21.3 (3.92) | 18.6 (1.51) |
B | 14.4 (2.97) | 22.2 (0.78) | 16.6 (4.22) | 20.9 (1.19) | |
C | 19.7 (3.27) | 20.2 (1.27) | 19.9 (3.02) | 18.1 (1.26) | |
2016 | A | 15.2 (2.47) | 14.6 (1.49) | 13.5 (1.39) | 17.6 (0.34) |
B | 14.9 (0.46) | 14.0 (1.27) | 17.4 (0.57) | 16.2 (1.02) | |
C | 16.7 (1.99) | 17.4 (1.07) | 16.9 (1.70) | 17.4 (2.22) | |
2017 | A | 10.6 (1.33) | 11.0 (0.64) | 12.0 (0.61) | 13.2 (0.97) |
B | 11.2 (0.38) | 13.7 (0.62) | 11.2 (2.03) | 13.0 (0.99) | |
C | 12.4 (1.66) | 12.2 (2.45) | 12.2 (1.60) | 12.0 (0.82) | |
2018 | A | 9.7 (1.85) | 9.4 (1.50) | 11.3 (0.93) | 14.8 (0.89) |
B | 11.4 (0.64) | 10.8 (0.39) | 12.5 (1.07) | 15.0 (0.49) | |
C | 14.7 (1.14) | 15.8 (0.41) | 15.2 (1.02) | 17.6 (1.03) | |
| ANOVA | df | F value | P value | Significance# |
2014 | iSPTC (S) | 2 | 24.423 | < 0.001 | *** |
Fertiliser type (F) | 3 | 3.045 | 0.050 | n.s. | |
S × F | 6 | 0.385 | 0.880 | n.s. | |
2015 | iSPTC (S) | 2 | 0.487 | 0.621 | n.s. |
Fertiliser type (F) | 3 | 6.693 | 0.002 | ** | |
S × F | 6 | 2.467 | 0.056 | n.s. | |
2016 | iSPTC (S) | 2 | 4.864 | 0.018 | * |
Fertiliser type (F) | 3 | 2.126 | 0.126 | n.s. | |
S × F | 6 | 2.274 | 0.074 | n.s. | |
2017 | iSPTC (S) | 2 | 0.739 | 0.489 | n.s. |
Fertiliser type (F) | 3 | 2.030 | 0.139 | n.s. | |
S × F | 6 | 1.929 | 0.121 | n.s. | |
2018 | iSPTC (S) | 2 | 60.049 | < 0.001 | *** |
Fertiliser type (F) | 3 | 26.890 | < 0.001 | *** | |
S × F | 6 | 1.866 | 0.132 | n.s. | |
#Significance of p-levels: n.s. = not significant (p > 0.05), * = 5 % (p ≤ 0.05), ** = 1 % (p ≤ 0.01), *** = 0.1 % (p ≤ 0.001). | |||||
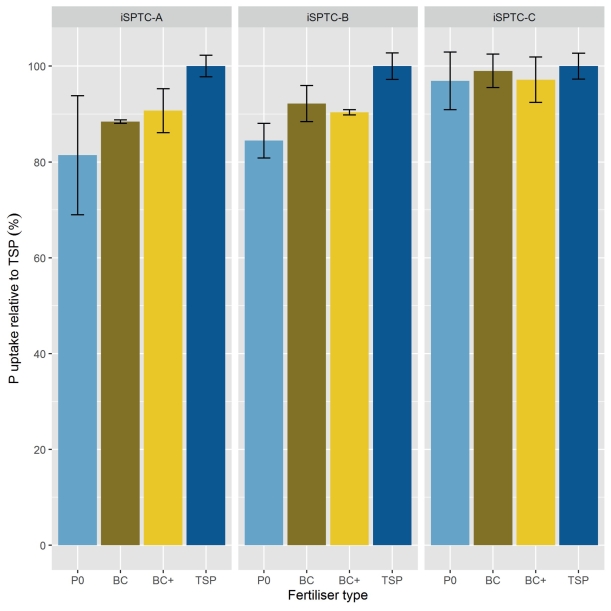
Fig. 2. Mean relative P uptake by grains with TSP = 100 % as affected by initial soil P-test class (iSPTC; A = severely deficient, B = deficient, C = sufficient) and type of P-fertiliser (P0, BC, BC+ (= BCplus), TSP) during the first crop rotation of the experiment from 2014–2018.
The total P uptake by grains, cumulated for the whole crop rotation, ranged between 67.6 kg ha–1 for the unfertilised, initially severely P-deficient soil and 87.8 kg ha–1 for the initially P-sufficient soil supplied with TSP fertiliser (Fig. 3), leading to a P-use efficiency of 30 to 39 % (225 kg ha–1 P applied in five years). A one-factorial analysis of variance of the total P uptake by grains cumulated for one crop rotation showed that soils with sufficiently available P concentrations were not affected by the fertiliser treatments (Fig. 3). In deficient and severely deficient soil, TSP fertilisation increased the P uptake by grain significantly in comparison to the control without P fertilisation, whilst the BC and BCplus fertilisation did not significantly increase the P uptake (Fig. 3). The total P uptake by grain of all years showed significant effects of the iSPTC (P value < 0.0001) as well as of the fertiliser treatments (P value = 0.0005) but no statistical evidence of any interaction of iSPTC and applied fertiliser type (P value = 0.3597) when analysed by a two-factorial analysis of variance.
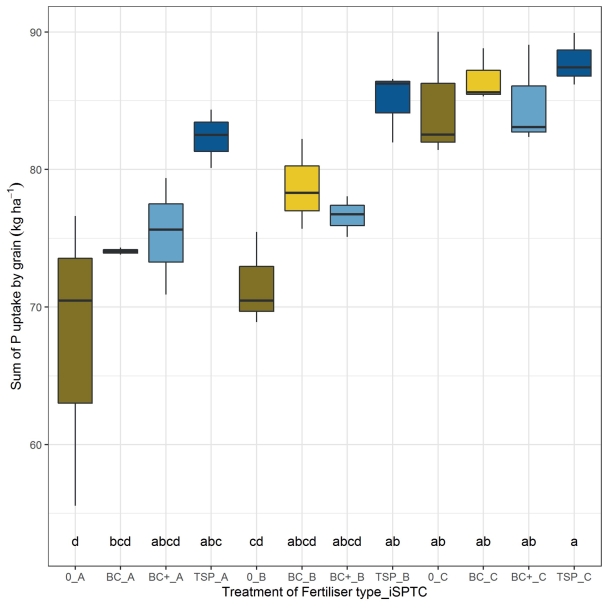
Fig. 3. Sum of P uptake by grain as affected by initial soil P-test class (iSPTC; A = severely deficient, B = deficient, C = sufficient) and type of P-fertiliser (P0, BC, BC+ (= BCplus), TSP) during the first crop rotation of the experiment from 2014–2018.
These results were reflected by the very low ANR results for the iSPTC-C of 0.9–1.4 % (Table 8). The ANR values were larger in soils that had lower initial P levels (iSPTC-A and -B). Both experimental factors influenced the ANR significantly but no statistical evidence of any interaction of iSPTC and applied fertiliser type were found (Table 8). Except for the control treatment without P fertilisation, in which soil P was depleted, all fertiliser applications resulted in a surplus in the P budget (Table 9). The P surplus was highest in treatments with the lowest P uptake and vice versa (Table 9).
Table 8. Apparent nutrient recovery (ANR) of grain as affected by initial soil P-test class (iSPTC; A = severely deficient, B = deficient, C = sufficient) and type of P-fertiliser (P0, BC, BCplus, TSP) during the first crop rotation of the experiment from 2014–2018 (means of three replicates and standard deviation in brackets)
Effect | Apparent nutrient recovery [%] | |||
P0 | BC | BCplus | TSP | |
iSPTC |
|
|
|
|
A | – | 2.9 (0.12) | 3.4 (1.89) | 6.6 (0.94) |
B | – | 3.2 (1.46) | 2.2 (0.66) | 5.9 (1.14) |
C | – | 0.9 (0.87) | 0.1 (1.63) | 1.4 (0.85) |
ANOVA | df | F value | P value | Significance# |
iSPTC (S) | 2 | 20.874 | < 0.001 | *** |
Fertiliser type (F) | 3 | 12.509 | < 0.001 | *** |
S × F | 6 | 1.526 | 0.242 | n.s. |
#Significance of p-levels: n.s. = not significant (p > 0.05), * = 5 % (p ≤ 0.05), ** = 1 % (p ≤ 0.01), *** = 0.1 % (p ≤ 0.001). | ||||
Table 9. P budget of grain as affected by initial soil P-test class (iSPTC; A = severely deficient, B = deficient, C = sufficient) and type of P-fertiliser (P0, BC, BCplus, TSP) during the first crop rotation of the experiment from 2014–2018 (means of three replicates and standard deviation in brackets)
Effect | P budget [kg ha–1] | |||
P0 | BC | BCplus | TSP | |
iSPTC |
|
|
|
|
A | –68 (10.8) | 151 (0.3) | 150 (4.2) | 143 (2.1) |
B | –72 (3.4) | 146 (3.3) | 148 (1.5) | 140 (2.6) |
C | –85 (4.7) | 138 (1.9) | 140 (3.7) | 137 (1.9) |
ANOVA | df | F value | P value | Significance# |
iSPTC (S) | 2 | 21.419 | < 0.001 | *** |
Fertiliser type (F) | 3 | 5779.945 | < 0.001 | *** |
S × F | 6 | 1.166 | 0.360 | n.s. |
#Significance of p-levels: n.s. = not significant (p > 0.05), * = 5 % (p ≤ 0.05), ** = 1 % (p ≤ 0.01), *** = 0.1 % (p ≤ 0.001). | ||||
Annual P fertiliser application (45 kg P ha–1) was much higher than the P removal by harvested products ranging from 8 to 24 kg P ha–1 depending on experimental treatments and crops grown (Table 5). Differences in the concentrations of plant available P (PCAL; concentrations 2018 minus concentrations 2013) were detected during the crop rotation (Table 10) which did not correlate with the P budget (r2 = 0.1501). The PCAL concentrations were significantly different between the iSPTCs and the applied fertiliser type but there was no evidence of any interaction of both factors (Table 10). Highly water-soluble TSP fertiliser increased the PCAL concentrations in all three iSPTCs, whereas the BC treatment led to a decrease in iSPTC-B and -C and the BCplus treatment to a decrease in iSPTC-C (Table 10). Only the application of TSP led to a change in soil P-test classes: iSPTC-A changed to soil P-test class B after two and iSPTC-B changed to soil P-test class C after four fertiliser applications. Similar effects were observed for the water-extractable P concentration in top soil after harvest, which increased for all three iSPTCs after five years of TSP application but decreased for all other treatments except for iSPTC-B and BCplus fertilisation (Table 11).
Table 10. Change (2018–2013, pre-experiment) of CAL-soluble P in top soil (0–30 cm) of experimental plots as affected by initial soil P-test class (iSPTC; A = severely deficient, B = deficient, C = sufficient) and type of P-fertiliser (P0, BC, BCplus, TSP) during the first crop rotation of the experiment from 2014–2018 (means of three replicates and standard deviation in brackets)
Effect | PCAL difference 2018–2013 [mg kg–1] | |||
P0 | BC | BCplus | TSP | |
iSPTC |
|
|
|
|
A | –4.0 (1.00) | 0.6 (0.25) | 1.8 (5.17) | 11.1 (2.25) |
B | –2.3 (2.68) | –2.8 (4.52) | 3.6 (0.59) | 13.3 (8.84) |
C | –12.9 (3.20) | –12.4 (1.79) | –8.5 (7.09) | 6.7 (11.25) |
ANOVA | df | F value | P value | Significance# |
iSPTC (S) | 2 | 12.000 | < 0.001 | *** |
Fertiliser type (F) | 3 | 17.370 | < 0.001 | *** |
S × F | 6 | 0.430 | 0.851 | n.s. |
#Significance of p-levels: n.s. = not significant (p > 0.05), * = 5 % (p ≤ 0.05), ** = 1 % (p ≤ 0.01), *** = 0.1 % (p ≤ 0.001). | ||||
Table 11. Change (2018–2013, pre-experiment) of water-soluble P in top soil (0–30 cm) of experimental plots as affected by initial soil P-test class (iSPTC; A = severely deficient, B = deficient, C = sufficient) and type of P-fertiliser (P0, BC, BCplus, TSP) during the first crop rotation of the experiment from 2014–2018 (means of three replicates and standard deviation in brackets)
Effect | Pwater difference 2018–2013 [mg kg–1] | |||
P0 | BC | BCplus | TSP | |
iSPTC |
|
|
|
|
A | –1.4 (0.30) | –0.2 (0.56) | –0.3 (1.27) | 2.1 (1.18) |
B | –0.9 (0.23) | –1.4 (0.16) | 0.4 (0.23) | 1.4 (0.70) |
C | –3.1 (0.77) | –3.0 (0.60) | –3.4 (2.03) | 0.7 (3.58) |
ANOVA | df | F value | P value | Significance# |
iSPTC (S) | 2 | 9.591 | 0.001 | ** |
Fertiliser type (F) | 3 | 9.793 | < 0.001 | *** |
S × F | 6 | 0.832 | 0.558 | n.s. |
#Significance of p-levels: n.s. = not significant (p > 0.05), * = 5 % (p ≤ 0.05), ** = 1 % (p ≤ 0.01), *** = 0.1 % (p ≤ 0.001). | ||||
Varying yield responses to P fertilisation in single years, as could be seen in the presented experiment (Table 6), are commonly reported (Lester et al., 2008) and can be explained by the low effects of freshly applied P fertilisers on P uptake and yield (Sylvester-Bradley et al., 2019). With ANR values below 7 %, almost all P taken up by plants was gained from the plant available P resources of the soil, confirming observations made in long-term P fertilisation experiments before (Merbach et al., 2010). Explaining also that statistically significant yield reductions of control plots (P0) were not observed, even though yield in iSPTC-A decreased markedly (Fig. 1). In iSPTC-C all tested fertilisers were able to maintain the mean relative yield (Fig. 1) during the first crop rotation, proving that yield can be sustained with any but also without (P0) fertiliser on soils with a sufficient plant available P status (Johnston & Poulton, 2019). Additional experimental years will allow the evaluation if BC and BCplus will be able to maintain yield levels considering that the PCAL and Pwater concentrations were reduced during the first crop rotation (Table 10 and 11). Mean ANRs of 0.1–1.4 % in iSPTC-C were very low confirming that applied P has only implications for soils with deficient plant available P status (Valkama et al., 2011). Higher mean ANRs of 2.2–6.6 % in iSPTC-A and -B support these findings and also that the overall efficiency to utilise inorganic P fertilisers by plants is generally less than 10 % (Baligar et al., 2001). The lower mean ANR for BC and BCplus in comparison to TSP points towards a lower agronomic efficiency of BCs. Nonetheless, in P deficient soils about 3 % of the applied P was utilised. According to Johnston & Poulton (2019) ANRs of fertilisers often range between 5 and 10 %. Only TSP fertilisation on P deficient soils achieved such ANRs, whilst all other fertiliser type/iSPTC combinations resulted in ANRs below 4 % (Table 8), which were in the order of values reported in a recent study by Sylvester-Bradley et al. (2019) about mineral fertiliser applications. The low ANR values are in contrast to findings of a pot experiment conducted with the same fertilisers resulting in much higher ANRs of < 3 %, 10–15 %, and > 18 % for BC, BCplus and TSP, respectively (Zimmer et al., 2019). These higher values can be attributed to limited soil that can be rooted in pot experiments constraining also the possibility of plants to use more distant soil P resources as well as to the controlled environment of glasshouse studies. It might also indicate that in the field experiment factors others than nutrient availability limited the crop yields during the study period. Exceptional weather conditions in some years were most likely reducing ANR values. A wet summer in 2017 postponed the harvest and thus induced most probably yield loss. The following year was very dry and hot in late spring and summer placing water constraints on plant growth. Furthermore, it can be assumed that the transformation from grassland to arable land in 2013 influenced e.g. mineralisation processes during the first experimental years. Therefore, the question if BCs have long-lasting effects for supplying P to crops cannot be answered conclusively after the first crop rotation of this long-term field experiment.
In spring, P uptake under plant available P deficient field conditions (iSPTC-A and -B) tended to increase in BCplus compared with BC treated plots (Table 4). However, BCplus did not lead to higher yields (Table 6) or P uptake at harvest (Table 7). This disagrees with results of a pot experiment conducted with the same fertilisers, reporting a statistically significant increase in P uptake through the application of BCplus (Zimmer et al., 2019). Then again, yields of the pot and field experiment were similar, with an increase by BCplus application in comparison to BC, however, these effects could not be confirmed statistically (Zimmer et al., 2019). In iSPTC-C, BC and BCplus reduced PCAL concentrations after one crop rotation. This reduction was lower in the BCplus than BC treatment, which had similar effects as the P0 treatment, suggesting a trend towards a better P solubility in BCplus treated soils with sufficient plant available P status. It also points towards an “in situ digestion” effect by the S component of BCplus (Fan et al., 2002). The initial question, if additional S components of BCplus can increase the plant availability of P in soil, yield, and P uptake by plants, can be affirmed only partly. In 2015, significantly higher P uptake by grains of winter oilseed rape in the BC fertilised plots compared with BCplus, TSP and P0 were unexpected, since oilseed rape does not form mycorrhizal symbiosis for P acquisition and its root system architecture is hampered by a low P environment (Yuan et al., 2016). However, Leppin (2007) showed that oilseed rape was able to access P from fertilised apatite (soft ground rock phosphate) in pot experiments, confirming findings of Hoffland et al. (1989). These authors showed that oilseed rape, as a reaction to P-starvation, was able to acidify the local rhizosphere by exudation of organic acids, which is a likely possibility to solubilise P (Hoffland et al., 1989; Leppin, 2007).
Until today, statistically significant interactions between iSPTC and fertiliser type of Pwater in spring or changes in PCAL and Pwater after harvest could not be established. Since pyrolysis of BC and BCplus above 800°C mainly creates insoluble HA, solubilisation of P was expected to be slow. However, whilst in iSPTC-A both bone char treatments resulted in only minor changes in the PCAL status of soils after harvest, BC fertiliser applied to iSPTC-B had a negative effect and BCplus a positive effect on the PCAL concentration (Table 10). Nevertheless, the BCs contribution to P uptake of grains at P deficient soils (Table 7, Fig. 3) indicates that the routine soil P-tests do not cover the complete pool of plant available P during a growing season. Exceptional high variation of the sum of P uptake by grain of the P0 treatment on iSPTC-A soil might also prevent statistical evidence for the severely P deficient soil (Fig. 3).The question, whether the initial P status of experimental soil affects the P solubility of BC and BCplus, needs further evaluation.
Generally, water extractable soil P correlates well with P uptake by plants (Zehetner et al., 2018) but is also used as an indicator for potential P leaching loss from soils (Wang et al., 2015). In spring, water extractable P concentrations were marginally higher in the BCplus than in the BC treated soil (Table 5) indicating a slightly better P supply to crops by BCplus. Due to the overall low levels of water extractable P concentrations for BC and BCplus, especially in P deficient soils of iSPTC-A and -B, a higher risk of P leaching losses cannot be expected.
The first crop rotation of a newly established field experiment testing the agronomic efficiency of BC and BCplus in comparison to highly water-soluble TSP fertiliser and P0 showed that in P deficient soils only TSP was able to increase concentrations of CAL-soluble P. In soils with sufficient plant available P status, no yield response of any fertiliser treatment was observed. At the same time, however, treatment effects on P uptake and soil PCAL concentrations of all fertilisers could be established in P deficient soils, even though for BCs only about half in size of the TSP treatment. The decrease of water- and CAL- soluble soil P of soils with sufficient P status reduces the risk of P losses into other ecosystems but it still needs to be proven that BC and/or BCplus will be able to maintain sufficient P supply for crops. In that case, BCs could be a sustainable P resource in the future; especially for P deficient soils, e.g. as a result of organic management without application of mineral P fertiliser or other P sources. Advantages of BCplus in comparison to BC were mainly seen during the period of major plant growth in spring. The reactions, by which the addition of sulfur in BCplus increases the P solubility and improves P uptake by crops, need further evaluation.
The authors would like to thank Dr. Karen Baumann and the two anonymous reviewers for their valued comments on earlier versions of this manuscript and Gudrun Gebensleben, Marina Kotschnew, and Frank Przebierala for their ongoing technical support. This project was funded in the frame of the Bioeconomy 2030 initiative of the Federal Ministry for Education and Research (BMBF; call: BonaRes; project InnoSoilPhos: No 031A558G and 031B0509A), research data will be made available on the BonaRes Data Portal.
The authors declare no conflicts of interest.
Baligar, V.C., N.K. Fageria, Z.L. He, 2001: Nutrient use efficiency in plants. Communications in Soil Science and Plant Analysis 32 (7-8), 921–950, DOI: 10.1081/css-100104098.
Buckwell, A., E. Nadeu, 2016: Nutrient recovery and reuse (NRR) in European agriculture: A review of the issues, opportunities, and actions, 1-96. Access: 21. September 2020, URL: https://www.nutrientplatform.org/wp-content/uploads/2016/09/Nutrient-Recovery-and-Reuse-in-European-Agriculture.pdf.
Cascarosa, E., G. Gea, J. Arauzo, 2012: Thermochemical processing of meat and bone meal: A review. Renewable and Sustainable Energy Reviews 16 (1), 942–957, DOI: 10.1016/j.rser.2011.09.015.
Cordell, D., J.-O. Drangert, S. White, 2009: The story of phosphorus: Global food security and food for thought. Global Environmental Change 19 (2), 292–305, DOI: 10.1016/j.gloenvcha.2008.10.009.
Eivazi, F., M.A. Tabatabai, 1977: Phosphatases in soils. Soil Biology and Biochemistry 9 (3), 167–172, DOI: 10.1016/0038-0717(77)90070-0.
EU, 2014: On the review of the list of critical raw materials for the EU and the implementation of the Raw Materials Initiative. Access: 03. November 2020, URL: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:52014DC0297&from=EN.
Fan, X., L. Habib, J. Fleckenstein, S. Haneklaus, E. Schnug, 2002: “In situ digestion” a concept to manage soil phosphate in organic farming. 13th International Fertilizer Symposium, 10-13 June 2002, Tokat, Turkey, 219-228.
Hoffland, E., G.R. Findenegg, J.A. Nelemans, 1989: Solubilization of rock phosphate by rape. Plant and Soil 113 (2), 161–165, DOI: 10.1007/BF02280176.
IUSS Working Group WRB, 2014: World reference base for soil resources 2014: International soil classification system for naming soils and creating legends for soil maps. Rome, FAO. Access: 03. November 2020, URL: http://www.fao.org/3/i3794en/I3794en.pdf.
Johnston, A.E., P.R. Poulton, 2019: Phosphorus in Agriculture: A Review of Results from 175 Years of Research at Rothamsted, UK. Journal of Environmental Quality 48 (5), 1133–1144, DOI: 10.2134/jeq2019.02.0078.
Kleinman, P.A., A. Sharpley, R. McDowell, D. Flaten, A. Buda, L. Tao, L. Bergstrom, Q. Zhu, 2011: Managing agricultural phosphorus for water quality protection: principles for progress. Plant and Soil 349 (1-2), 169–182, DOI: 10.1007/s11104-011-0832-9.
Leppin, T., 2007: Mobilisierungspotential unterschiedlicher Pflanzen für stabile Phosphatformen im Boden. PhD Thesis, Justus-Liebig-Universität Gießen, 1-132.
Lester, D.W., C.J. Birch, C.W. Dowling, 2008: Fertiliser N and P applications on two Vertosols in north-eastern Australia. 1. Comparative grain yield responses for two different cultivation ages. Australian Journal of Agricultural Research 59 (3), 247–259, DOI: 10.1071/AR07169.
Meier, U., 2018: Growth stages of mono- and dicotyledonous plants: BBCH Monograph. Open Agrar Repositorium, DOI: 10.5073/20180906-074619.
Merbach, W., A. Deubel, A. Gransee, S. Ruppel, A.-K. Klamroth, 2010: Phosphorus solubilization in the rhizosphere and its possible importance to determine phosphate plant availability in soil. A review with main emphasis on German results. Archives of Agronomy and Soil Science 56 (2), 119–138. DOI: 10.1080/03650340903005640.
Morshedizad, M., P. Leinweber, D. Zimmer, 2016: Effect of bone chars on phosphorus-cadmium-interactions as evaluated by three extraction procedures. Journal of Plant Nutrition and Soil Science 179, 388–398. DOI: 10.1002/jpln.20150060.
Murphy, J., J.P. Riley, 1962: A modified single solution method for determination of phosphate in natural waters. Analytica Chimica Acta 26 (1), 31–36. DOI: 10.1016/S0003-2670(00)88444-5.
Neset, T.-S.S., D. Cordell, 2012: Global phosphorus scarcity: identifying synergies for a sustainable future. Journal of the Science of Food and Agriculture 92 (1), 2–6, DOI: 10.1002/jsfa.4650.
Postma, J., F. Clematis, E.H. Nijhuis, E. Someus, 2013: Efficacy of four phosphate-mobilizing bacteria applied with an animal bone charcoal formulation in controlling Pythium aphanidermatum and Fusarium oxysporum f.sp. radicis lycopersici in tomato. Biological Control 67 (2), 284–291, DOI: 10.1016/j.biocontrol.2013.07.002.
Schinner, F., E. Kandeler, R. Öhlinger, R. Margesin (Eds.), 1996: Methods in soil biology. Berlin, Heidelberg, Springer-Verlag.
Scholz, R.W., F.-W. Wellmer, 2013: Approaching a dynamic view on the availability of mineral resources: What we may learn from the case of phosphorus? Global Environmental Change 23 (1), 11–27, DOI: 10.1016/j.gloenvcha.2012.10.013.
Schüller, H., 1969: Die CAL-Methode, eine neue Methode zur Bestimmung des pflanzenverfügbaren Phosphates in Böden. Zeitschrift für Pflanzenernährung und Bodenkunde 123 (1), 48–63, DOI: 10.1002/jpln.19691230106.
Siebers, N., F. Godlinski, P. Leinweber, 2012: The phosphorus fertilizer value of bone char for potatoes, wheat, and onions: first results. Landbauforschung 62 (1-2), 59-64.
Siebers, N., F. Godlinski, P. Leinweber, 2014: Bone char as phosphorus fertilizer involved in cadmium immobilization in lettuce, wheat, and potato cropping. Journal of Plant Nutrition and Soil Science 177 (1), 75–83, DOI: 10.1002/jpln.201300113.
Smith, D.R., M.L. Macrae, P.J.A. Kleinman, H.P. Jarvie, K.W. King, R.B. Bryant, 2019: The latitudes, attitudes, and platitudes of watershed phosphorus management in North America. Journal of Environmental Quality 48 (5), 1176–1190, DOI: 10.2134/jeq2019.03.0136.
Sylvester-Bradley, R., A. Rollett, E. Downing, S. Dudman, M. Slater, N. Morris, S. Knight, P. Withers, 2019: Cost-effective Phosphorus Management on UK Arable Farms: Includes the report on Work-Package 3: Improving the efficiency of fresh P applications, 66 p. Access: 03. November 2020, URL: https://ahdb.org.uk/cost-effective-phosphorus-management-on-uk-arable-farms-sustainable-p.
Tabatabai, M.A., J.M. Bremner, 1969: Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biology and Biochemistry 1 (4), 301–307, DOI: 10.1016/0038-0717(69)90012-1.
Ulén, B., M. Bechmann, L. Øygarden, K. Kyllmar, 2012: Soil erosion in Nordic countries – future challenges and research needs. Acta Agriculturae Scandinavica, Section B - Soil & Plant Science 62 (sup2), 176–184, DOI: 10.1080/09064710.2012.712862.
Valkama, E., R. Uusitalo, E. Turtola, 2011: Yield response models to phosphorus application: a research synthesis of Finnish field trials to optimize fertilizer P use of cereals. Nutrient Cycling in Agroecosystems 91 (1), 1–15, DOI: 10.1007/s10705-011-9434-4.
van der Paauw, F., H.A. Sissingh, J. Ris, 1971: Een verbeterde methode van fosfaatextractie van grond met water: het Pw-getal. Verslagen van Landbouwkundige Onderzoekingen 749, 54-63.
van Dijk, K.C., J.P. Lesschen, O. Oenema, 2016: Phosphorus flows and balances of the European Union Member States. Science of The Total Environment 542, Part B, 1078–1093, DOI: 10.1016/j.scitotenv.2015.08.048.
Vassilev, N., E. Martos, G. Mendes, V. Martos, M. Vassileva, 2013: Biochar of animal origin: a sustainable solution to the global problem of high-grade rock phosphate scarcity? Journal of the Science of Food and Agriculture 93 (8), 1799–1804, DOI: 10.1002/jsfa.6130.
VDLUFA, 1991: Methodenbuch I Die Untersuchung von Böden: Bestimmung von Schwermetallen im Aufschluss mit Königswasser, A 2.4.3.1 (A 2.4.3.1).
VDLUFA, 2000: Methodenbuch II.2 Die Untersuchung von Sekundärrohstoffdüngern, Kultursubstraten und Bodenhilfsstoffen: Bestimmung des Gesamtphosphors: Aufschluss mit Königswasser, 3.2.1.3 (3.2.1.3).
Vogeler, I., J. Rogasik, U. Funder, K. Panten, E. Schnug, 2009: Effect of tillage systems and P-fertilization on soil physical and chemical properties, crop yield and nutrient uptake. Soil and Tillage Research 103 (1), 137–143. DOI: 10.1016/j.still.2008.10.004.
Wang, Y.T., T.Q. Zhang, I.P. O’Halloran, Q.C. Hu, C.S. Tan, D. Speranzini, I. Macdonald, G. Patterson, 2015: Agronomic and environmental soil phosphorus tests for predicting potential phosphorus loss from Ontario soils. Geoderma 241–242 (0), 51–58, DOI: 10.1016/j.geoderma.2014.11.001.
Warren, G., J. Robinson, E. Someus, 2009: Dissolution of phosphorus from animal bone char in 12 soils. Nutrient Cycling in Agroecosystems 84 (2), 167–178. DOI: 10.1007/s10705-008-9235-6.
Wiesler, F., T. Appel, K. Dittert, T. Ebertseder, T. Müller, L. Nätscher, H.-W. Olfs, M. Rex, K. Schweitzer, D. Steffens, F. Taube, W. Zorn, 2018: Phosphordüngung nach Bodenuntersuchung und Pflanzenbedarf: VDLUFA-Standpunkt. Access: 03. November 2020, URL: https://www.vdlufa.de/Dokumente/Veroeffentlichungen/Standpunkte/2018_Standpunkt_P-Duengung.pdf.
Withers, P.J.A., P.M. Haygarth, 2007: Agriculture, phosphorus and eutrophication: a European perspective. Soil Use and Management 23, 1–4, DOI: 10.1111/j.1475-2743.2007.00116.x.
Yuan, P., G.-D. Ding, H.-M. Cai, K.-M. Jin, M.R. Broadley, F.-S. Xu, L. Shi, 2016: A novel Brassica-rhizotron system to unravel the dynamic changes in root system architecture of oilseed rape under phosphorus deficiency. Annals of Botany 118 (2), 173–184, DOI: 10.1093/aob/mcw083.
Zehetner, F., R. Wuenscher, R. Peticzka, H. Unterfrauner, 2018: Correlation of extractable soil phosphorus (P) with plant P uptake: 14 extraction methods applied to 50 agricultural soils from Central Europe. Plant, Soil and Environment 64 (No. 4), 192–201, DOI: 10.17221/70/2018-PSE.
Zimmer, D., J. Kruse, N. Siebers, K. Panten, C. Oelschläger, M. Warkentin, Y. Hu, L. Zuin, P. Leinweber, 2018: Bone char vs. S-enriched bone char: Multi-method characterization of bone chars and their transformation in soil. Science of The Total Environment 643, 145–156, DOI: 10.1016/j.scitotenv.2018.06.076.
Zimmer, D., K. Panten, M. Frank, A. Springer, P. Leinweber, 2019: Sulfur-enriched bone char as alternative P fertilizer: spectroscopic, wet chemical, and yield response evaluation. Agriculture 9 (1), 21, DOI: 10.3390/agriculture9010021.
Zwetsloot, M.J., J. Lehmann, T. Bauerle, S. Vanek, R. Hestrin, A. Nigussie, 2016: Phosphorus availability from bone char in a P-fixing soil influenced by root-mycorrhizae-biochar interactions. Plant and Soil 408 (1), 95–105, DOI: 10.1007/s11104-016-2905-2.
Table S1. Potential acidic phosphatase activity in spring 2016 – 2018 as affected by initial soil P-test classes (iSPTC; A = severely deficient, B = deficient, C = sufficient) and type of P-fertiliser (P0, BC, BCplus, TSP) (means of three replicates and standard deviation in brackets)
Year | iSPTC | Potential acidic phosphatase activity [μg p-NP g–1 h–1] | |||
P0 | BC | BCplus | TSP | ||
2016 | A | 285 (24.1) | 301 (28.1) | 254 (39.3) | 248 (27.8) |
B | 266 (32.1) | 284 (21.4) | 265 (1.6) | 268 (13.7) | |
C | 269 (21.2) | 263 (29.2) | 284 (38.3) | 260 (16.6) | |
2017 | A | 301 (35.3) | 340 (48.9) | 299 (15.2) | 283 (9.4) |
B | 303 (32.3) | 321 (19.4) | 288 (9.2) | 294 (14.4) | |
C | 308 (43.8) | 275 (14.7) | 302 (44.7) | 288 (29.5) | |
2018 | A | 251 (44.1) | 266 (26.9) | 254 (12.7) | 225 (7.1) |
B | 247 (22.1) | 252 (9.6) | 262 (26.3) | 244 (24.5) | |
C | 247 (22.3) | 254 (29.2) | 262 (26.3) | 249 (24.4) | |
| ANOVA | df | F value | P value | Significance# |
2016 | iSPTC (S) | 2 | 0.067 | 0.936 | n.s. |
Fertiliser type (F) | 3 | 1.704 | 0.195 | n.s. | |
S × F | 6 | 1.460 | 0.238 | n.s. | |
2017 | iSPTC (S) | 2 | 0.659 | 0.528 | n.s. |
Fertiliser type (F) | 3 | 1.256 | 0.314 | n.s. | |
S × F | 6 | 1.470 | 0.234 | n.s. | |
2018 | iSPTC (S) | 2 | 0.072 | 0.930 | n.s. |
Fertiliser type (F) | 3 | 1.226 | 0.324 | n.s. | |
S × F | 6 | 0.356 | 0.899 | n.s. | |
#Significance of p-levels: n.s. = not significant (p > 0.05), * = 5 % (p ≤ 0.05), ** = 1 % (p ≤ 0.01), *** = 0.1 % (p ≤ 0.001). | |||||
Table S2. Potential alkaline phosphatase activity in spring 2016 – 2018 as affected by initial soil P-test classes (iSPTC; A = severely deficient, B = deficient, C = sufficient) and type of P-fertiliser (P0, BC, BCplus, TSP) (means of three replicates and standard deviation in brackets)
Year | iSPTC | Potential alkaline phosphatase activity [μg p-NP g–1 h–1] | |||
P0 | BC | BCplus | TSP | ||
2016 | A | 69 (13.0) | 74 (10.0) | 57 (12.4) | 69 (14.3) |
B | 76 (19.0) | 64 (11.8) | 68 (13.4) | 55 (2.5) | |
C | 70 (14.3) | 70 (7.3) | 72 (23.1) | 68 (5.1) | |
2017 | A | 73 (5.6) | 66 (11.7) | 73 (15.5) | 72 (3.8) |
B | 84 (5.7) | 70 (8.0) | 79 (1.4) | 72 (12.8) | |
C | 72 (12.2) | 75 (2.6) | 65 (9.4) | 70 (10.2) | |
2018 | A | 74 (8.5) | 76 (11.7) | 75 (2.2) | 83 (4.8) |
B | 87 (16.2) | 69 (19.9) | 79 (11.1) | 79 (8.3) | |
C | 78 (13.3) | 82 (18.2) | 69 (12.7) | 79 (5.5) | |
| ANOVA | df | F value | P value | Significance# |
2016 | iSPTC (S) | 2 | 0.340 | 0.715 | n.s. |
Fertiliser type (F) | 3 | 0.674 | 0.577 | n.s. | |
S × F | 6 | 0.843 | 0.551 | n.s. | |
2017 | iSPTC (S) | 2 | 1.548 | 0.235 | n.s. |
Fertiliser type (F) | 3 | 0.566 | 0.566 | n.s. | |
S × F | 6 | 0.783 | 0.593 | n.s. | |
2018 | iSPTC (S) | 2 | 0.069 | 0.934 | n.s. |
Fertiliser type (F) | 3 | 0.476 | 0.702 | n.s. | |
S × F | 6 | 0.738 | 0.625 | n.s. | |
#Significance of p-levels: n.s. = not significant (p > 0.05), * = 5 % (p ≤ 0.05), ** = 1 % (p ≤ 0.01), *** = 0.1 % (p ≤ 0.001). | |||||