

First report on Pythium myriotylum as pathogen on duckweed (Lemna minor L.) in hydroponic systems in Germany
Erstnachweis von Pythium myriotylum als Krankheitserreger an Wasserlinsen (Lemna minor L.) in hydroponischen Systemen in Deutschland
Journal für Kulturpflanzen, 73 (9-10). S. 316–323, 2021, ISSN 1867-0911, DOI: 10.5073/JfK.2021.09-10.02, Verlag Eugen Ulmer KG, Stuttgart
 | This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/deed.en). Dies ist ein Open-Access-Artikel, der unter den Bedingungen der Creative Commons Namensnennung 4.0 International Lizenz (CC BY 4.0) zur Verfügung gestellt wird (https://creativecommons.org/licenses/by/4.0/deed.de). |
Duckweed is a promising resource for future feed and food production as well as wastewater treatment. However, diseases and pests can critically limit the performance of the production systems. Patches of discolored and bleached duckweed (Lemna minor L.) appeared in hydroponic systems and spread rapidly through the crop. Pythium myriotylum was confirmed as the causing pathogen by microbiological and molecular biological analysis. This is the first report of P. myriotylum on duckweed in Germany. The result and possible countermeasures are discussed.
Key words: Hydroponics, plant disease, discoloration, bleaching, control, water lentils, Lemnaceae
Wasserlinsen sind eine vielversprechende Ressource für die zukünftige Produktion von Futter- und Nahrungsmitteln sowie die Abwasserreinigung. Krankheiten und Schädlinge können die Leistung der Produktionssysteme jedoch kritisch einschränken. In hydroponischen Systemen traten Flecken missfarbiger und gebleichter Wasserlinsen (Lemna minor L.) auf, die sich rasch im Bestand ausbreiteten. Durch mikrobiologische und molekularbiologische Untersuchungen wurde Pythium myriotylum als ursächlicher Schaderreger eindeutig nachgewiesen. Dies ist der erste Bericht über P. myriotylum an Wasserlinsen in Deutschland. Das Ergebnis und mögliche Gegenmaßnahmen werden erörtert.
Stichwörter: Hydroponik, Pflanzenkrankheit, Verfärbung, Bleichung, Gegenmaßnahmen, Entengrütze, Lemnaceae
Due to high biomass production, nutritional values and easy digestibility, duckweeds, also referred to as water lentils, are of great interest for food and feed production. Duckweeds of several genera including Lemna are suitable for human as well as animal consumption and cultivated in hydroponic or aquaponic systems (Somerville et al., 2014; Appenroth et al., 2017; Chakrabarti et al., 2018). In addition, duckweed can be used for removing nutrients and pollutants during wastewater treatment (Paterson, 2017; Iatrou et al., 2019). Thus, duckweed production might provide valuable contributions to solving current and future food, agricultural as well as environmental problems (Leng, 1999).
Duckweed fronds are multiplying nearly exponentially in clonal growth (Landolt, 1986) thus the biomass can be harvested continuously (Cheng & Stomp, 2009). Main limiting factors for growth and harvest are nutrient supply and space (Driever et al., 2005; Lasfar et al., 2007). However, the development of duckweed can be limited by algal growth (Roijackers et al., 2004). Furthermore, as in other crop production systems, diseases and pests can critically limit the performance of hydroponic or aquaponic systems (Folorunso et al., 2020) and thus jeopardize the duckweed production success. However, few pathogens are known for duckweed such as Tracya lemnae, Olpidium amoebae (syn. Reesia amoeboides) and Pythium spp., but information on pathogenicity is scarce (Brandenburger, 1985; Rejmankova et al., 1986). According to the sparse literature, Pythium species such as P. aphanidermatum and P. myriotylum seem most relevant for duckweed production (Rejmankova et al., 1986; Flaishman et al., 1997).
In July 2020, rapidly growing patches of discolored, and eventually bleached fronds of Lemna minor L. (clone 9441; Germany) were observed (Fig. 1) in experimental hydroponic production systems for duckweed at the University of Applied Sciences Osnabrück, Germany. The first signs of the disease appeared 7 days after the start of cultivation in pools with no water movement, hence no duckweed movement (Fig. 1A). In the following six days, one specific spot (Fig. 1B) grew from 3 cm to 26 cm in diameter. In a circulatory hydroponic system, mainly in zones with very low duckweed movement, spots were observed 15 days after the start of cultivation (Fig. 1 C). In both system types water temperatures ranged between 24 to 27°C.

Fig. 1. Patches of discolored and bleached fronds of Lemna minor in pools of hydroponic systems. A: standing water, diseased (left) and healthy (right), B: standing water, single patch in detail, C: closed circulatory system, single patch in detail.
In order to be able to take appropriate countermeasures, inhibit further spread and prevent repeated occurrence, the cause of this disease was investigated phytopathologically.
Samples of healthy and diseased Lemna minor L. (clone 9441; Germany) fronds were send by mail in a water containing plastic flask to the lab of the plant protection service, Oldenburg, Lower Saxony, Germany. Fronds were transferred to glass dishes filled with tap water to a height of about 2 cm and examined visually under a binocular (Nikon SMZ800).
Without pretreatment, individual discolored or bleached duckweed fronds were laid out on half strength potato dextrose agar (PDA50%; 19,5 g l–1 potato extract glucose agar [Carl Roth, Karlsruhe, Germany], 7,5 g l–1 agar) and on carrot piece agar (CPA: 50 g l–1 grated carrot, 15 g l–1 agar) and incubated at room temperature. Pure isolates were produced, transferring tips of single hyphae to PDA50% and CPA.
Resulting mycelium was examined microscopically (Nikon Eclipse Ni). Microscopic images and measurements of characteristic structures were made using the Nikon DS-Fi3 camera and the imaging software NIS-Elements D 5.20.01. For each relevant structure, 100 measurements were made. Measurements are reported as (minimum–)mean ± standard deviation (–maximum) and as median.
To determine growth characteristics, plugs were taken from fully grown PDA50% and CPA plates of a selected isolate using a cork borer (5 mm diameter) and transferred centrally to fresh PDA50% and CPA, respectively. Incubation was performed at five temperature levels (20, 25, 30, 35, 40°C) in the dark. The diameter of the colonies (n = 5) was measured at 24 h and 48 h.
In order to prove pathogenicity of the isolated strain, five clear 500 ml plastic beakers with lid were filled about 1 cm high with nutrient solution (N-Medium) according to Appenroth (2015). Plugs with 13 mm diameter (produced as mentioned above) were submerged (5/beaker) and healthy Lemna minor (50 plantlets/beaker) were placed on the water surface. Incubation was at 30°C under light (d/n 16/8 h). A control, treated the same way but not inoculated with the isolated strain, was run in parallel. Diseased duckweeds were subject for re-isolation in order to fulfill Koch’s postulates.
For DNA isolation freshly grown mycelium on one PDA plate was scraped off in a 2 mL reaction tube and slightly homogenized with a pestle prior to further processing. DNA was extracted using the innuPREP Plant DNA Kit (Analytik Jena AG, Jena, Germany) following the manufacturer’s instructions given in protocol 1. The mitochondrially encoded gene cytochrome c oxidase subunit I (COI) was partially amplified using the primer pair OomCoxI-Levup and OomCoxI-Levlo (Robideau et al., 2011).
PCR was performed in a reaction volume of 50 μl containing final concentrations of 1x ready-to-use MyTaq Plant-PCR Mix (Bioline Meridian Bioscience, London, UK), 5 μl genomic DNA (1:10 diluted) and 0.4 μM of each primer. Reaction volume was brought up to 50 μl with sterile ddH20.
Amplification conditions were 2 min at 95°C, followed by 35 cycles at 95°C for 1 min, 55°C for 1 min, 72°C for 1 min, followed by a final extension at 72°C for 10 min. PCR products were purified using innuPREP PCRpure Kit (Analytik Jena AG, Jena, Germany). For bidirectional Sanger sequencing OomCoxI-Levup and OomCoxI-Levlo were used as well. A consensus sequences was prepared by using DNA Sequence Assembler v5 (Heracle BioSoft S.R.L, Arges, Romania). The sequence of the re-isolated strain was gained in the same manner and aligned with the original sequence.
The sequence of the initial isolate was deposited in GenBank (https://www.ncbi.nlm.nih.gov/Genbank/) under the accession number MW679676.
GenBank was searched for similar sequences using blastn (Altschul et al., 1990) for preliminary species identification.
Detailed species identification was performed by generating a data set for Pythium Clade B according to the reference material of Robideau et al. (2011). Phylogenetic analysis was performed by using MEGA X (Kumar et al., 2018). The sequences were aligned by using the incorporated software MUSCLE (Edgar, 2004). Leading and trailing gaps were cut to a total of 680 positions per sequence. Maximum likelihood interference was done using the Tamura-Nei model (Tamura & Nei, 1993) for substitution and 1000 bootstrap replicates.
During examination of bleached duckweed fronds under binoculars, hyphae were regularly observed, which were recognized as unseptate under the microscope. Therefore, an assignment to the oomycetes seemed probable.
Isolation on both media was successful. Mycelium appearing macro- and microscopically identical grew out of almost all transferred duckweed fronds.
Microscopically, unseptate mycelium with irregularly shaped, elongated appressoria, filamentous and inflated sporangia, smooth oogonia [(19,7–)27,0 ± 2,4(–31,9) μm, median 27,3 μm; n = 100] and aplerotic oospores [(15,8–)22,4 ± 1,3(–27,8) μm, median 22,4 μm; n = 100] formed in high numbers were recognized as structures relevant for identification (Fig. 2). Chlamydospores were not observed. Due to these characteristics, Pythium was recognized as genus. Following the key to the species of Plaats-Niterink (1981), the isolate was determined as P. myriotylum.
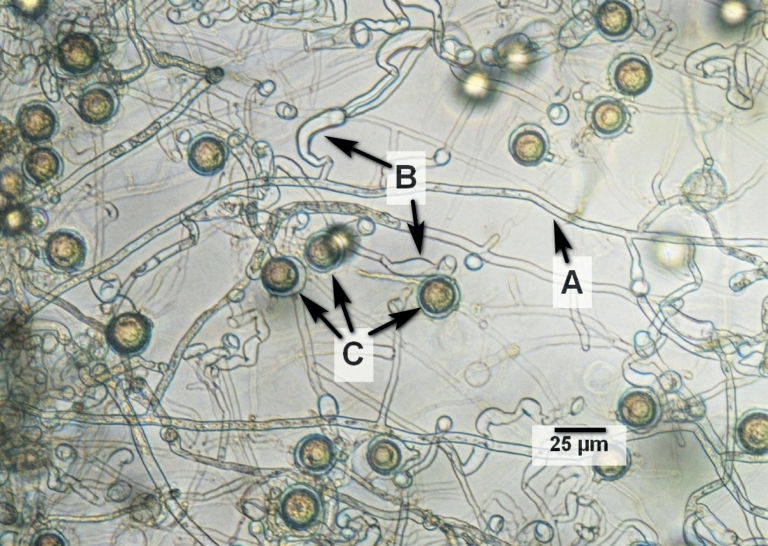
Fig. 2. Microscopic image of the isolate from Lemna minor on PDA50%. A: unseptate hyphae, B: irregularly shaped, elongated appressoria, C: aplerotic oospores.
On PDA50%, the isolate grew dense, uniform and somewhat fluffy without special structure (Fig. 3A), whereas the colonies on CPA appeared much looser, branched, and rough (Fig. 3B).
In the temperature range studied (20–40°C), the isolate grew well at all selected levels (Fig. 4). At 20°C it showed the lowest growth rate, but growth increased with the temperature rising up to 35°C on both media, although growth rate was generally higher on CPA compared to PDA50%. The data indicate that the optimum for growth can be assumed to be around 35°C. Even at 40°C, a considerable growth rate could be observed, indicating a mesophilic species with a maximum cardinal temperature above 40°C.
![Fig. 4. Average diameter [mm] of Pythium myriotylum colonies (n = 5) isolated from Lemna minor at different temperatures after 24 and 48 hours growth on CPA and PDA50%.
Bars indicate standard deviation. Growth limited to 85 mm by the diameter of the Petri dish.](bilder/jfk_2021_09-10_brand_et_al_bld-004.jpg)
Fig. 4. Average diameter [mm] of Pythium myriotylum colonies (n = 5) isolated from Lemna minor at different temperatures after 24 and 48 hours growth on CPA and PDA50%. Bars indicate standard deviation. Growth limited to 85 mm by the diameter of the Petri dish.
One day after inoculation first bleached duckweed fronds were observed. Already after two days at 30°C, almost all of the duckweeds in the inoculated beakers showed discoloration and bleaching as previously observed, whereas the plantlets in the non-inoculated control thrived without symptoms.
From the diseased duckweeds, a Pythium could be re-isolated and determined to be P. myriotylum by morphological means.
The COI sequences of the initial isolate and the re-isolation were similar. The sequence was 100% identical to the voucher specimen of P. myriotylum and P. zingiberis. In the phylogenetic tree it clustered with these two species (Fig. 5). No further resolution could be achieved by molecular means.
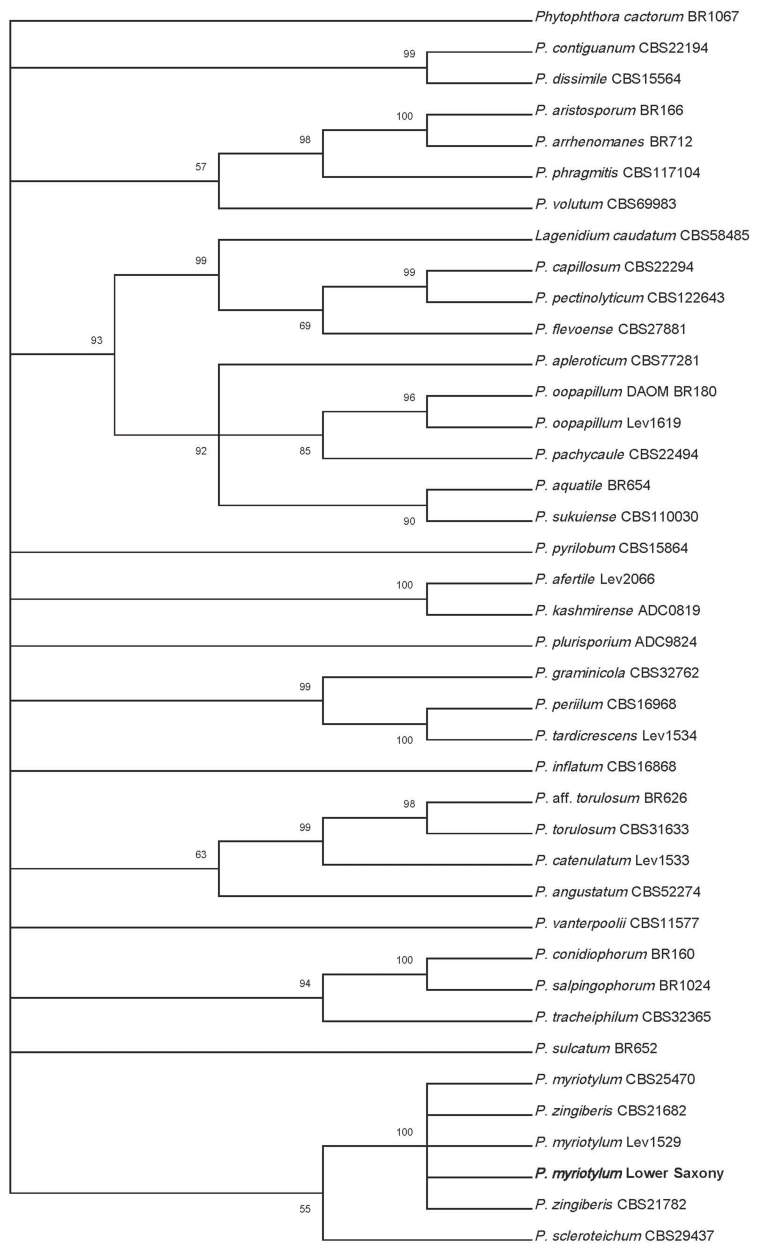
Fig. 5. Phylogenetic reconstruction (Maximum Likelihood) based on partial cytochrome oxidase c subunit I (COI) sequences of Pythium Clade B according to Robideau et al. (2011) including the sequence of the Pythium isolated from Lemna minor in Lower Saxony (bold). The bootstrap consensus tree is inferred from 1000 replicates. Number on branches denote bootstrap support values for maximum likelihood analyses. Only supported values > 50% are displayed. The tree was rooted to Phytophthora cactorum.
The results of both microbiological and molecular biological investigations consistently show that the pathogen belongs to the genus Pythium. Pythium species are plant pathogens regarded as soilborne with broad host spectrum infecting mainly seedlings as well as roots and stem bases, causing damping-off and rot, respectively (Plaats-Niterink, 1981). However, propagules of Pythium and other oomycetes are easily dispersed with water and regularly found in natural waters (Czeczuga et al., 2005), aquaponic systems (Folorunso et al., 2020), and irrigation systems of horticulture (Hoitink et al., 1991; Bush et al., 2003; Sutton et al., 2006; Ivors & Moorman, 2017).
Although Pythium is commonly found in these aquatic habitats, specific data of disease development in duckweed are scarce (Rejmankova et al., 1986; Flaishman et al., 1997). On the one hand, this may be due to the actually rare occurrence of diseases caused by biological, physical or chemical factors that suppress Pythium (Postma et al., 2000). On the other hand, investigations may have been omitted due to the low importance of duckweed as crop so far. This could change in the future, with expanded use and increased interest, and the number of observations of corresponding diseases could rise.
The identification on species level to P. myriotylum by means of sequencing is supported by the determined morphological characteristics as well as the growth rate and temperature preference of the isolate, which are in good accordance with the literature (Plaats-Niterink, 1981; Kröber, 1985; Rejmankova et al., 1986; Le et al., 2015). However, differentiation between some closely related Pythium species is difficult both morphologically, physiologically, and molecularly due to intraspecific variability, considerable influence of environmental factors on morphological features and high degree of genetic similarities (Plaats-Niterink, 1981; Kröber, 1985; Perneel et al., 2006; Le et al., 2017a, 2017b). With respect to P. myriotylum, a reliable differentiation from P. zingiberis is not possible by methods currently employed to determine Pythium species (Robideau et al., 2011; Le et al., 2015, 2017a, 2017b). Therefore, Le et al. (2017a) propose to include P. zingiberis in P. myriotylum, as taxonomic rules require prioritization of this name. We share this approach and therefore consider the isolate obtained from L. minor as P. myriotylum.
Worldwide distributed, especially in the tropics and subtropics, P. myriotylum causes severe losses in several important agricultural and horticultural crops and ornamentals from different plant families (Plaats-Niterink, 1981; Wang et al., 2003; Perneel et al., 2006). The pathogen was found in greenhouse soilless cultivation systems on different plant hosts (Anderson et al., 1997; Hong et al., 2004; Pantelides et al., 2017; Vitale et al., 2018). Furthermore, P. myriotylum is pathogenic to aquatic plants in natural and artificial waters (Rejmankova et al., 1986; Czeczuga et al., 2005) and a strain of P. myriotylum “from duckweed” is listed in the catalogue of the American Type Culture Collection (Anonymus, 2021). Flaishman et al. (1997) report on the occurrence of a disease caused by this pathogen in production tanks of Lemna gibba observing similar symptoms as described above.
As Koch’s postulates are fulfilled within this work, it can be safely stated that in the investigated case P. myriotylum is the causal agent for the observed disease of L. minor. To our knowledge, this is the first report on P. myriotylum on L. minor in Germany.
Pests and diseases can critically limit plant production, even in hydroponic or aquaponic systems, so integrated plant protection measures are necessary to achieve the set goals (Folorunso et al., 2020). In principle, hygiene measures that prevent pests from reaching the host plant are of high importance for plant protection. To prevent Pythium from being introduced into production systems, plants must be free of infestation at the start of cultivation and the water must be pathogen-free or sterilised (Hong et al., 2017). Apart from these main entry points, Pythium can also be transmitted by dust (Sánchez et al., 2001), meaning that in more or less open cultivation systems, whether in a greenhouse or outdoors, entry can hardly be permanently prevented. This might be more feasible in closed biotechnological facilities with high general hygienic standard.
The key factor for infection by P. myriotylum and disease development is high temperature (Plaats-Niterink, 1981; Rejmankova et al., 1986; Flaishman et al., 1997; Wang et al., 2003). As shown by Rejmankova et al. (1986) duckweeds are able to overgrow the infection when temperature is below 22°C. With increasing temperatures, duckweed population development is more and more affected by the disease. Similar observations were reported from Fortnum et al. (2000) for the infection of P. myriotylum during the production of tobacco seedlings in float systems. Therefore, optimized temperature control in production systems is crucial to prevent an outbreak and severe progression of the disease.
Another major factor in disease development is plant density (Rejmankova et al., 1986; Flaishman et al., 1997), suggesting that adapted production management helps prevent the disease. Whether early and generous removal of infested duckweed blotches is sufficient for sustainable control must be tested. In this respect, our observation that bleached patches first appeared in areas with stagnant or very slow-flowing water may be an indication that higher flow rates can reduce the likelihood of the disease occurring. Further research would be useful in this context.
Regarding biological control options, bacteria and fungi antagonistically active against P. myriotylum are known from terrestrial systems (Mbarga et al., 2012; Jimtha et al., 2016). Furthermore, antagonistic microorganisms can have significant influence on the activity of Pythium and the development of caused root rots in hydroponic systems (Paulitz, 1997; Postma et al., 2000; Sutton et al., 2006). It might be assumed that similar relationships also affect the development of diseases in general and outbreaks of P. myriotylum in hydroponic duckweed production in particular, however, no data are available.
Physical methods (e.g., UV irradiation, heating, sonication, filtration) can be used to minimize the likelihood of transmission in hydroponic systems (Hong et al., 2017; Mori & Smith, 2019). Because physical disinfection acts only at one point in the system and has no lasting effect, transmission can be prevented between subunits of hydroponic systems, but not spread within a subunit (Hong et al., 2017).
The use of chemical substances active against pathogens in hydroponic systems such as chlorine, chlorine dioxide, copper, ozone or approved plant protection products must be critically evaluated. Questions need to be addressed regarding the management of risks to workers and the environment, food and feed safety, compatibility for crops, and development of resistance (Hong et al., 2017).
We are indebted to Lucija Jahn, Manal Morad and Jenny Rebentisch for technical support.
The authors declare that there are no conflicts of interest.
Altschul, S.F., W. Gish, W. Miller, E.W. Myers, D.J. Lipman, 1990: Basic local alignment search tool. Journal of Molecular Biology 215, 403-410.
Anderson, M.G., B.A. Fortnum, S.B. Martin, 1997: First Report of Pythium myriotylum in a Tobacco Seedling Float System in South Carolina. Plant Disease 81 (2), 227, DOI: 10.1094/PDIS.1997.81.2.227D.
Anonymus, 2021: Pythium myriotylum (ATCC® 42401™). Access: 28.01.2021. URL: https://www.lgcstandards-atcc.org/~/ps/42401.ashx.
Appenroth, K.-J., 2015: Media for in vitro-cultivation of duckweed. Duckweed Forum 3, 180-186.
Appenroth, K.-J., K.S. Sree, V. Böhm, S. Hammann, W. Vetter, M. Leiterer, G. Jahreis, 2017: Nutritional value of duckweeds (Lemnaceae) as human food. Food Chemistry 217, 266-273, DOI: 10.1016/j.foodchem.2016.08.116.
Brandenburger, W., 1985: Parasitische Pilze an Gefäßpflanzen in Europa. Stuttgart, Gustav Fischer Verlag.
Bush, E.A., C.X. Hong, E.L. Stromberg, 2003: Fluctuations of Phytophthora and Pythium spp. in components of a recycling irrigation system. Plant Disease 87 (12), 1500-1506, DOI: 10.1094/PDIS.2003.87.12.1500.
Chakrabarti, R., W.D. Clark, J.G. Sharma, R.K. Goswami, A.K. Shrivastav, D.R. Tocher, 2018: Mass production of Lemna minor and its amino acid and fatty acid profiles. Frontiers in Chemistry 6 (479), 1-6, DOI: 10.3389/fchem.2018.00479.
Cheng, J., A.M. Stomp, 2009: Growing Duckweed to Recover Nutrients from Wastewaters and for Production of Fuel Ethanol and Animal Feed. CLEAN - Soil, Air, Water 37 (1), 17–26, DOI: 10.1002/clen.200800210.
Czeczuga, B., A. Godlewska, B. Kiziewicz, E. Muszyńska, B. Mazalska, 2005: Effect of Aquatic plants on the abundance of aquatic zoosporic fungus species. Polish Journal of Environmental Studies 14 (2), 149-158.
Driever S.M., E.H. van Nes, R.M.M. Roijackers, 2005: Growth limitation of Lemna minor due to high plant density. Aquatic Botany 81 (3), 245-251, DOI: 10.1016/j.aquabot.2004.12.002.
Edgar, R.C., 2004: MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32 (5), 1792-1797, DOI: 10.1093/nar/gkh340.
Flaishman, M.A., E. Hadar, E. Hallak-Herr, 1997: First report of Pythium myriotylum on Lemna gibba in Israel. Plant Disease 81 (5), 550, DOI: 10.1094/PDIS.1997.81.5.550C.
Folorunso, E.A., K. Roy, R. Gebauer, A. Bohatá, J. Mraz, 2020: Integrated pest and disease management in aquaponics: A metadata-based review. Reviews in Aquaculture, 1-25, DOI: 10.1111/raq.12508.
Fortnum, B.A., J. Rideout, S.B. Martin, D. Gooden, 2000: Nutrient solution temperature affects Pythium root rot of tobacco in greenhouse float systems. Plant Disease 84 (3), 289-294, DOI: 10.1094/pdis.2000.84.3.289.
Hong, S.Y., J.-W.Kim, Y.K. Kang, Y.M. Yang, Y.S. Kang, 2004: Potato basal stem rot caused by Pythium myriotylum in hydroponic cultural systems. Research in Plant Disease 10 (1), 13-16, DOI: 10.5423/RPD.2004.10.1.013.
Hong, C., G.W. Moorman, W. Wohanka, C. Büttner (Eds.), 2017: Biology, Detection, and Management of Plant Pathogens in Irrigation Water, St. Paul, USA, APS Press.
Hoitink, H.A.J., R.P. Fynn, R.W. McMahon, A. Atmatjidou, 1991: Transmission of plant pathogens in an ebb and flood system. Ohio Florists Association Bulletin 742, 5-9.
Iatrou, E.I., E. Kora, A.S. Stasinakis, 2019: Investigation of biomass production, crude protein and starch content in laboratory wastewater treatment systems planted with Lemna minor and Lemna gibba. Environmental Technology 40 (20), 2649-2656, DOI: 10.1080/09593330.2018.1448002.
Ivors, K.L., G.W. Moorman, 2017: Oomycete plant pathogens in irrigation water. In: Biology, Detection, and Management of Plant Pathogens in Irrigation Water. Hong, C., G.W. Moorman, W. Wohanka, C. Büttner (Eds.), St. Paul, USA, APS Press, 57-64.
Jimtha, J.C, P. Jishma, G.B. Arathy, C. Anisha, E.K. Radhakrishnan, 2016: Identification of plant growth promoting rhizosphere Bacillus sp. WG4 antagonistic to Pythium myriotylum and its enhanced antifungal effect in association with Trichoderma. Journal of Soil Science and Plant Nutrition 16 (3), 578-590, DOI: 10.4067/S0718-95162016005000026.
Kröber, H., 1985: Erfahrungen mit Phytophthora de Bary und Pythium Pringsheim. Mitteilungen aus der Biologischen Bundesanstalt für Land- und Forstwirtschaft Berlin-Dahlem 225. Berlin und Hamburg, Paul Parey.
Kumar, S., G. Stecher, M. Li, C. Knyaz, C., K. Tamura, 2018: MEGA X: Molecular evolutionary genetics analysis across computing platforms. Molecular Biology and Evolution 35 (6), 1547-1549, DOI: 10.1093/molbev/msy096.
Landolt, E., 1986: Biosystematic investigations in the family of duckweeds (Lemnacae) (vol. 2) – The family of Lemnacae – a monographic study vol. 1. Veröffentlichungen des Geobotanischen Institutes der Eidg. Technischen Hochschule, Stiftung Rübel in Zürich 71..
Lasfar, S., F. Monette, L. Millette, A. Azzouz, 2007: Intrinsic growth rate: a new approach to evaluate the effects of temperature, photoperiod and phosphorus-nitrogen concentrations on duckweed growth under controlled eutrophication. Water Research 41 (11), 2333–2340, DOI: 10.1016/j.watres.2007.01.059.
Le, D.P., E.A.B. Aitken, M.K. Smith, 2015: Comparison of host range and pathogenicity of isolates of Pythium myriotylum and Pythium zingiberis. Acta Horticulturae: 1105, DOI: 10.17660/ActaHortic.2015.1105.1.
Le, D.P., M.K. Smith, E.A.B. Aitken, 2017a: Species delimitation in Pythium species complexes: the case of Pythium myriotylum Drechsler and Pythium zingiberis Takahashi. Mycological Progress 16 (3), 257-267, DOI: 10.1007/s11557-017-1272-6.
Le, D.P., M.K. Smith, E.A.B. Aitken, 2017b: Genetic variation in Pythium myriotylum based on SNP typing and development of a PCR-RFLP detection of isolates recovered from Pythium soft rot ginger. Letters in Applied Microbiology 65 (4), 319-326, DOI: 10.1111/lam.12779.
Leng, R.A., 1999: Duckweed – a tiny aquatic plant with enormous potential for agriculture and environment. Rome, FAO.
Mbarga, J.B., G. Martijn ten Hoopen, J. Kuaté, A. Adiobo, M.E.L. Ngonkeu, Z. Ambang, A. Akoa, P.R. Tondje, B.A.D. Begoude, 2012: Trichoderma asperellum: A potential biocontrol agent for Pythium myriotylum, causal agent of cocoyam (Xanthosoma sagittifolium) root rot disease in Cameroon. Crop Protection 36, 18-22, DOI: 10.1016/j.cropro.2012.02.004.
Mori, J., R. Smith, 2019: Transmission of waterborne fish and plant pathogens in aquaponics and their control with physical disinfection and filtration: A systematized review. Aquaculture 504, 380-395, DOI: 10.1016/j.aquaculture.2019.02.009.
Pantelides, I.S., M.-D. Tsolakidou, A. Chrysargyris, N. Tzortzakis, 2017: First report of root rot of hydroponically grown peppermint (Mentha × piperita) caused by a Pythium myriotylum in Cyprus. Plant Disease 101 (9), 1682, DOI: 10.1094/PDIS-02-17-0250-PDN.
Paterson, J., 2017: Removing phosphate from wastewater: evaluation of the performance of duckweed (Lemna minor) operating under cool temperate conditions. PhD thesis, University of Leeds, 214 p.
Paulitz, T.C., 1997: Biological control of root pathogens in soilless and hydroponic systems. Hortscience 32 (2), 193-196.
Perneel, M., J.T. Tambong, A. Adiobo, C. Floren, F. Saborío, A. Lévesque, M. Höfte, 2006: Intraspecific variability of Pythium myriotylum isolated from cocoyam and other host crops. Mycological Research 110 (5), 583-593, DOI: 10.1016/j.mycres.2005.12.002.
Plaats-Niterink, A.J. van der, 1981: Monograph of the genus Pythium. Studies in Mycology 21. Baarn, Centraalbureau voor Schimmelcultures.
Postma, J., M.J.E.I.M. Willemsen-de Klein, J.D. van Elsas, 2000: Effect of indigenous microflora on the development of root and crown rot caused by Pythium aphanidermatum in cucumber grown in rockwool. Phytopathology 90 (2), 125-133, DOI: 10.1094/PHYTO.2000.90.2.125.
Rejmankova, E., M. Blackwell, D.D. Culley, 1986: Dynamics of fungal infections in duckweeds (Lemnaceae). Veröffentlichungen des Geobotanischen Institutes der Eidg. Technischen Hochschule, Stiftung Rübel in Zürich 87, 178-189.
Robideau, G.P., A.W.A.M. de Cock, M.D. Coffey, H. Voglmayr, H. Brouwer, K. Bala, D.W. Chitty, N. Désaulniers, Q.A. Eggertson, C.M.M. Gachon, C.-H. Hu, F.C. Küpper, T.L. Rintoul, E. Sarhan, E.C.P. Verstappen, Y. Zhang, P.J.M. Bonants, J.B. Ristaino, C.A. Lévesque, 2011: DNA barcoding of oomycetes with cytochrome c oxidase subunit I and internal transcribed spacer. Molecular Ecology Resources 11 (6), 1002-1011, DOI: 10.1111/j.1755-0998.2011.03041.x.
Roijackers, R., S. Szabó, M. Scheffer, 2004: Experimental analysis of the competition between algae and duckweed. Archiv für Hydrobiologie 160 (3), 401–412, DOI: 10.1127/0003-9136/2004/0160-0401.
Sánchez, J., J.S. Olivares, E. Gallego, 2001: Occurrence and pathogenicity of Pythium spp. in the dust deposited on the greenhouse roofs in the Poniente region of Almeria (south-east Spain). Journal of Plant Pathology 83 (1): 13-19.
Somerville, C., M. Cohen, E. Pantanella, A. Stankus, A. Lovatelli, 2014: Small-scale aquaponics food production. FAO Fisheries and Agriculture Technical Paper 589. Rome, FAO.
Sutton, J.C., C.R. Sopher, T.N. Owen-Going, W. Liu, B. Grodzinski, J.C. Hall, R.L. Benchimol, 2006: Etiology and epidemiology of Pythium root rot in hydroponic crops: current knowledge and perspectives. Summa Phytopathologica, 32 (4), 307-321, DOI: 10.1590/S0100-54052006000400001.
Tamura, K., M. Nei, 1993: Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Molecular Biology and Evolution 10, 512-526.
Vitale, S., L. Luongo, E. Marinelli, A. Belisario, 2018: First report of Pythium myriotylum as a causal agent of crown and root rot in soilless green bean cultivation in Italy. Plant Disease 102 (3), 688, DOI: 10.1094/PDIS-07-17-0942-PDN.
Wang, P.H., C.Y. Chung., Y.S. Lin, Y. Yeh, 2003: Use of polymerase chain reaction to detect the soft rot pathogen, Pythium myriotylum, in infected ginger rhizomes. Letters in Applied Microbiology 36, 116-120, DOI: 10.1046/j.1472-765x.2003.01272.x.