

Journal für Kulturpflanzen, 74 (03-04). S. 49–62, 2022 | DOI: 10.5073/JfK.2022.03-04.03 | Niemann et al.
Reducing initial aphid infestation by use of coloured mulch foils and newly developed biodegradable spray-films
Reduzierung des Initialbefalls durch Aphiden unter Nutzung von gefärbten Mulchfolien und neu entwickelten bioabbaubaren Sprühfilmen
 | (c) The author(s) 2022 This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/deed.en). |
Submitted/accepted for publication: 29 October 2021/11 March 2022 |
A newly invented biodegradable sprayable film based on renewable raw materials was modified in colour to reduce the landing and settlement of alate aphids (Macrosiphum euphorbiae, Nasonovia ribisnigri) on iceberg lettuce (Lactuca sativa L., var. crispa). The sprayable films were compared with green and black conventional PE-foils and the cultivation on bare soil. In field trials, migration and host settlement of insect pests were investigated with special consideration of colour contrasts and light reflection. The lettuce was evaluated for alate aphid infestation at the time of initial infestation and at the time of harvest. The aim was to reduce the initial infestation number with alate individuals and to minimize the demand for additional intervening measurements for plant protection. The black-grey spray film led to reduced numbers of aphids at initial infestation date. Evaluation at harvest time shows the need of secondary measurements for reliable pest control. The quality of the lettuce heads was not negatively affected by the spray treatment.
integrated pest management, currant-lettuce aphid, renewable resources, colour-contrast, wavelength-dependent behaviour
Eine neu entwickelte, biologisch abbaubare, sprühfähige Folie auf Basis nachwachsender Rohstoffe wurde farblich modifiziert, um den Zuflug und die Ansiedlung von Blattläusen (Macrosiphum euphorbiae, Nasonovia ribisnigri) auf Eisbergsalat (Lactuca sativa, var. crispa) zu reduzieren. Die sprühfähigen Folien wurden mit grünen und schwarzen konventionellen PE-Folien und dem Anbau auf Boden verglichen. In Feldversuchen wurden der Zuflug und die Ansiedlung von Schadinsekten unter besonderer Berücksichtigung von Farbkontrasten und Lichtreflexion untersucht. Der Salat wurde auf den Befall mit Blattläusen zum Zeitpunkt des Erstbefalls und zum Zeitpunkt der Ernte untersucht. Ziel war es, den Erstbefall mit Blattläusen zu reduzieren und den Bedarf an zusätzlichen Maßnahmen zum Pflanzenschutz zu minimieren. Der schwarzgraue Sprühfilm führte zu einer reduzierten Anzahl von Blattläusen zum Zeitpunkt des Erstbefalls. Die Auswertung zum Erntezeitpunkt zeigt die Notwendigkeit von zusätzlichen Maßnahmen für eine zuverlässige Schädlingsbekämpfung. Die Qualität der Salatköpfe wurde durch die Sprühfolien nicht negativ beeinflusst.
integrierter Pflanzenschutz, Johannisbeerblattlaus, nachwachsende Rohstoffe, Farbkontrast, wellenlängenabhängiges Verhalten
Aphids are one of the economically most important pest groups in the cultivation of plants. Since the population to be observed in the field is always based on immigrated individuals, control of initial infestation is the basis for restriction of population built up. Due to the exponential and fast growth of an aphid population, a large population quickly develops from single immigrated individuals.
The currant-lettuce aphid Nasonovia ribisnigri is the most common aphid species (Sternorrhyncha: Hemiptera) in lettuce cultivation (Reinink & Dieleman, 1993; Hommes et al., 2003) and is difficult to control with chemical and biological methods, especially due to its cryptic habitat in lettuce inner leaves. However, there are also populations with high resistance to various insecticides (Rufingier et al., 1997; Kift et al., 2004). It is known to infest more than 160 plant species from 15 different families (Holman, 2009), but there might be many more possible host plants. Primary hosts are Ribes ssp. only. Secondary hosts are much more diverse in the number of species, but in plant production only the infestation of Lactuca ssp. and Cichorium ssp. is of economic relevance. N. ribisnigri is found on secondary hosts on all parts of the plant, except flowers, but prefers young and/or soft tissues such as petals or young leaves. Therefore, when growing lettuces, monitoring at an advanced stage of lettuce development can only be carried out destructively and combating the aphids on the heartleaves with chemical, and even systemic insecticides is very difficult (Diaz et al., 2007). N. ribisnigri has damage potential on both the primary and secondary host. If there is no possibility of dispersion (e.g. in breeding), this also leads to the death of many secondary host species (own data, unpublished). Under practical conditions, on the primary hosts, the direct damaging effect of the sucking activity, which is limited to rolling of the leaves and growth depression, is relatively low. However, the aphids are able to act as vectors of the vein band mosaic virus of gooseberry (Blackman & Eastop, 2000). The direct damage to the secondary host lettuce, endive and chicory, is also low but still of economic importance. The secondary damage results from the increase of fungal infections caused by the honeydew secretion of the aphids inside the head. The wholesale and retail trade even in the case of a few aphids found in the product rejects lettuce batches.
Colours and colour contrasts have a strong influence on the behaviour of aphids. In immigrating individuals, green and yellow colour reflections trigger a landing reflex (Moericke, 1950; 1955; Döring & Spaethe, 2009), which is also controlled by light intensity (brightness) and the UV component of radiation (Döring & Chittka, 2007). However, exceptions prove the rule here, and are probably host plant dependent (Döring & Chittka, 2007; Straw et al., 2011; Farnier et al., 2014). For cultivated areas, it was shown that colonization of the contrasting borders from soil to plant at the edge of the field can be particularly strong (Hooks & Fereres, 2006). For example, green lettuce plants showed more aphid visits (by Myzus persicae and Macrosiphum euphorbiae) than reddish-brown lettuce plants (Müller, 1964), whose reflection values are closer to those of the soil. For aphids, it was shown that the contrast between plant and soil is important for targeted landing on the host plant and that landing rates can be reduced if the contrast is compensated by coloured (e.g. green) backgrounds (Döring et al., 2004; Hooks & Fereres, 2006; Döring, 2014). The decreasing attractiveness of potential aphid landing targets due to reduced contrast was already demonstrated with pan-trap experiments by Moericke (1955), Barro (1991), Döring (2014) and Döring & Röhrig (2016), where lower contrasts between traps and background also led to reduced number of aphids trapped, even when using attractive colours.
Green mulch has been shown to disturb aphid colonization within the field, but it can also increase visual plot localization by aphids from a distance, and thus immigration into the mulch field margins (Jones & Chapman, 1968; Hommes et al., 2003).
Reflective grey and silver foils showed a reduced immigration of aphids in many experiments, mostly resulting in increasing yields (Greer & Dole, 2003; Hommes et al., 2003; Shimoda, 2018). Unfortunately, the reflection spectrum of the foils used in these experiments is often missing, which makes it difficult to evaluate the results. Various grey/silver materials can have very different reflection spectra (Hommes et al., 2003; Döring et al., 2004). Another interfering factor is the use of traps and plants in the same experiment. The traps used for evaluation represent an additional colour stimulus (depending on colour) and/or contrast which may influence the landing behavior of the aphids.
The aim of the current paper was to compare a newly invented sprayable and biodegradable film with conventional practices (foil and bare soil) in deterring aphid landing in lettuce fields.
The sprayable films were compared to conventional culture practices like cultivation on bare soil and on green and black polyethylene (PE) foil (following named foil). If text refers to various materials for ground cover (film + foil), the term mulch is used. The hypothesis is that all coloured foils will reduce the initial infestation with aphids in contrast to cultivation on bare soil. In addition, the spray films should reduce the number of alighting aphids as strong as the PE foil will do.
The five different treatments used in the field experiment are shown in Table 1.
Table 1. Used mulch materials and supplier references for the five different treatments in the field experiment.
Treatment | Material | Supplier |
NF | Open soil without film/foil | - |
PEB | PE-foil Black | 25 my Premium-Foil, 1.2 m wide, Hartmann-Brockhaus, |
SFB | Spray Film Black-grey | own development, coloured with Black Pearls 800, Cabot |
PEG | PE-foil Green | 100 my foil, 1.5 m wide, Supporters GmbH, Lampertheim, Germany |
SFG | Spray Film Green | own development, coloured with Bentone French green soil and Italian gold ochre, Kremer Pigmente, Aichstetten, Germany |
The experiments were performed on an area of 80 m × 20 m. Each plot had a size of 8.5 m × 5.5 m, and the distance between two plots was 2 m in each direction. Within this area, iceberg lettuce (Lactuca sativa var. crispa) was planted (May 7th 2018) on 7.5 m × 4.5 m; the resulting edges of 0.5 m width were left free (treatment NF) or covered with foil like the cultivation area. The conventional foil strips were drawn in manually (May 7th 2018), and the sprayable film treatments were applied directly to the ground (May 6th 2018) with an airless sprayer (WagnerŠ Superfinish 7000) before planting. With a S10 527/235 nozzle (Art.nr. 271049, J.Wagner GmbH, Markdorf, Germany) at a pressure of approx. 200-220 bar, a quantity of 1.9 litres per square meter was applied.
All 15 experimental plots consisting of 3 dams were manually planted with 240 young lettuce plants each (12 rows with 20 plants each, with 4 rows per dam, planting distance 30 cm, row distance 30 cm, and distance of 30 cm to 40 cm between dams). Figure S5 shows one complete experimental plot (i.e. one replicate of a treatment) for all treatments. The number of replicates per treatment was three. Randomization was done for all 15 plots (a completely randomized design, with the restriction that replications of one treatment were not allowed to be located next to each other) and then filled in the field plan (Fig. S1). A blocked design was not possible because of limited field width. Note that in Figs. 3, S1 and S3 one row of plant symbols (circles) corresponds to the 4 rows of plants per dam. The numbering of rows and plants clarifies this, since indeed not all rows and plants of the three dams are shown in the heat map graphs.
For the evaluation, 30 plants per plot were assessed. For each plot, the 1st, 5th, 10th, 15th and 20th plants in the plant rows 1, 4, 5, 8, 9 and 12 were rated. This resulted in an evaluation raster of 0.9 m × 1.2 m in the field. Detailed graphical overview for the plot design (Figs. 3, S1 and S3) and pictures of foil and film materials used in the experiments (Fig. S5) are given in the supplementary. The raw data and statistical code for R are also provided online (Niemann et al., 2022).
Weeds were mechanically removed weekly to prevent an impact on aphid immigration. In the first non-destructive evaluation, a small mirror was used to inspect the underside of the leaves by visual counting of aphids at the beginning of the infestation on May 16. A second, destructive, evaluation was carried out at the time of harvest on June 22.
Preliminary studies (own study 2016, unpublished) showed that it takes a distance of around two meters between plot border and plot centre to be able to evaluate edge effects in lettuce. As field size was limited, each of the five treatments had three simultaneous replicates. Since another trial set in the same year and three in the year 2017 remained without any infestation by aphids, it was only possible to work with these data from 2018.
Reflection spectra for film/foil materials, dry soil and lettuce leaves were measured with the spectrophotometer Lambda 900 UV/VIS/NIR (Perkin-Elmer Instruments, Norwalk, USA) in order to draw possible correlations between the reflection of the ground surface and the immigration of aphids. Each film/foil and soil sample was measured three times at different randomly selected points on the sample surface and the mean was calculated. The reflection properties of plant leaves were measured 6 times; 3 times for the three youngest and oldest leaves, respectively (always upper side, central veins were excluded). Reflection spectra are shown in Fig. 1.
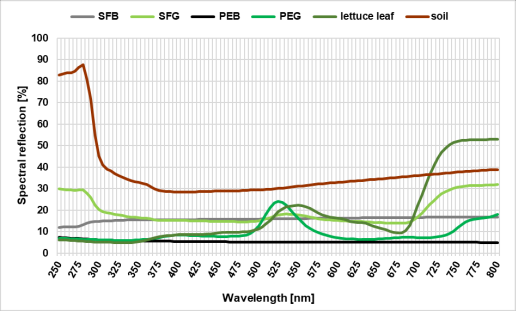
Fig. 1. Reflection spectra of the soil (NF), mulch-materials and the experimental plant Lactuca sativa, var. crispa. For abbreviations see Table 1.
The software R (R Core Team, 2019) was used for the statistical analysis. Data management was done using the tidyverse packages (Wickham, 2017). Since independence of the single observations (number of aphids per plant) within the subsamples (location) in each plot cannot be assumed, these observations were summed up separately for plants located in the centre of the plot (round dots in the heatmap, see Fig. 3) or on the border (squares in the heatmap). All further statistical analyses were run on these two sums per plot.
Since these sums depend on different numbers of lettuce plants (12 for the centre, 18 for the border) they were scaled to be (y + 1)/n. The shift of y + 1 was necessary to avoid zeros in the dependent variable, since the statistical model was run on logarithmized scaled sums as described below (and the logarithm of zero is not defined).
A linear mixed model using the lme4 package (Bates et al., 2015) was fitted in which the logarithmized scaled sums depend on the treatment, the location and their interaction as fixed effects and the plots as random effects.
Based on this model, an ANOVA was run and pairwise mean comparisons of the effects of the film/foil treatments were done using the emmeans package (Lenth, 2018). All tests were run with α=0.05. Please note that the comparisons of model-based means were run as differences on the log-link. Therefore, back transformation to the original scale yields ratios between these means. Graphics were made using the package ggplot2 (Wickham, 2017). The model described above was fitted to the total number of aphids as well as the number of alate aphids found in evaluation one (2018-05-16) and also to the total number of aphids found at the second evaluation (2018-06-22).
The R-code for modelling, ANOVA tables, model-based means of the scaled rates and their corresponding comparisons are given in the supplementary (Niemann et al., 2022).
The reflection spectra of the different treatments and the lettuce leaf are shown in Fig. 1. The “green” materials lettuce leaf and PEG showed typical peaks for bright green tones around 550 nm and lower reflection values in the blue (430 nm- 490 nm) and yellow (560 nm- 600 nm) range. SFG showed just a small increase at 550 nm but much higher reflection of all other wavelength than the two other “green” materials and a high reflection in the UV-range between 250 nm to 300 nm. PEB showed reflection values between 7% and 5%, SFB reflection ranges between 12% and 17%. The dry soil shows a high reflection in the UV range up to a peak at 280 nm (~ 90% refl.). From there, after a short fall to 30% at 385 nm, the curve flattens out sharply and then rises to around 40% at 800 nm.
Figure 2 shows the number of alate aphids found on the lettuce plants in the first evaluation for each treatment, separated into outer- (dots) and inner-plants (triangles) and as least square means with confidence interval, over all repetitions. Highest least square mean is shown by treatments PEG (0.294), SFG (0.291) and NF (0.257), PEB showed 0.110. The treatment SFB (0.068) was statistically different from all other treatments.
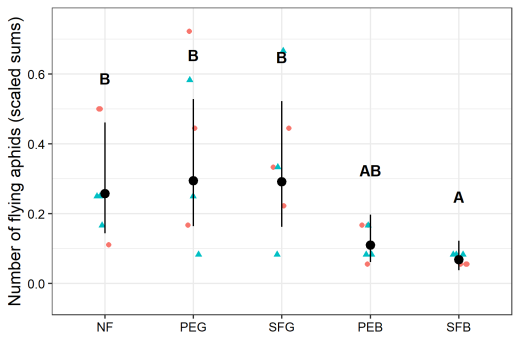
Fig. 2. Scaled sums of the total number of alate aphids per location in each plot (May 16, 2018). The sums were scaled to be (y + 1)/n. Dots indicate sums from the border plants, triangles indicate sums from the centre of a plot. The black dots are model-based least square means of the scaled sums and their 95% confidence intervals (black bars). The letters indicate significant differences (α=0.05) between logarithmized means.
Figure 3 shows the total number of aphids (N. ribisnigri, only 1 alate individual) for every evaluation plant by increasing intensity of the colour in the second evaluation. In all treatments, more alate aphids on outer plants than on plants in the centre of the plots were counted, but the ANOVA (Table S17) did not show statistical significance (p-value=0.067) for a location effect neither on average, nor within the plots (p-value = 0.91). The mean ratios of counted aphids between inner and outer plants for each treatment are given in Table S18. As for the total number of aphids, only the treatment effect was significant (p-value = 0.025, Table S14). The number of infested plants was 36 (40%, NF), 44 (49%, PEG), 17 (19%, SFG), 19 (21% PEB), 12 (13%, SFB).
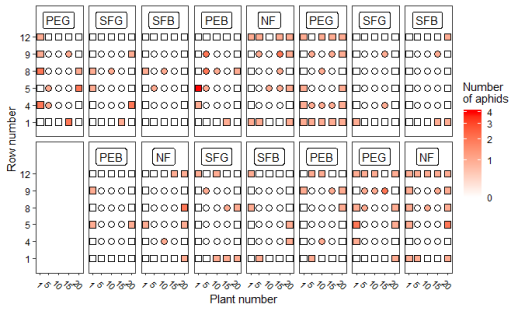
Fig. 3. Heatmap for the total number of aphids (June 22, 2018) with overview over all evaluated lettuce plants (n=450). Five treatments with three replications each on 15 randomized plots (n=30 per plot). Total number of aphids (alate and apterous) found in the 2nd evaluation is represented by increasing colour hue. Evaluated plants are indicated by plant number (x-axis) and row number (y-axis). Outer plants at the edge to bare soil are shown as square dots, plants surrounded by other plants as round dots. Orientation is north to south (from left to right).
In the field experiment, a significant effect was observed in the SFB treatment, which in the first evaluation showed almost no infestation by alate aphids (Fig. 2). Moreover, the SFB treatment also showed the lowest infestation of all treatments in the second evaluation (Fig. 3, Fig. S4) even if not significant.
Previous experiments by Jones & Chapman (1968) showed reduced virus spread in the combination of lettuce and black foil, and studies by Johnson et al. (1967) also showed reduced aphid infestation using black PE-foil, but the relevant effects were not discussed and involved effects remain unclear. However, spectrometer measurements of the PEB and the SFB treatment in our experiment (Fig. 1) did not show any remarkable reflection values indicating a repellent effect by reflected wavelengths (nm). Since the surface properties of the ground covers (film, foil, soil) were not in focus of this study and not varied independently we cannot prove it, but like Moericke (1955) and Döring et al. (2004) we assume an effect by those. The SFB and PEB treatment both showed a glossy optic (to human eye) in contrast to the other three treatments. For further experiments, we would recommend the measurement of the gloss with a gloss meter over the experimental period. In addition to the optical effects mentioned above, it is also possible that the different surface properties of the mulch materials influenced post-landing behaviour (Moericke, 1950; 1955; Finch & Collier, 2000; Döring et al., 2004), and may have led to a take-off by the aphids. Further research would be needed to determine where the effect is coming from.
The green treatments SFG and PEG were not able to reduce the initial infestation in our experiment. In addition, PEG also showed highest infestation in the second evaluation. This may result from the fact that we did not succeed in exactly reproducing the spectral green of the plants. Especially the SFG treatment shows lower reflection values as the lettuce and the overall curve is more like the one from the soil as from the lettuce. This could have resulted in infestation rates like the NF treatment has shown in Fig. 2 (and Fig. S4). If we compare the spectra of the lettuce plant with the green treatments, the lettuce shows a higher reflectance at 550 nm (yellow-green) as the other two treatments. For the PEG treatment this reflection might have acted like a trap in contrast to the green background and therefore helps the aphids to navigate correctly to the host plants. Experiments with green mulch materials showing increasing yellow hues could demonstrate this. Plot size could also have an influence on the migration rate. For further field experiments with coloured foils, we would recommend bigger plots and smaller number of treatments.
The significant difference between PEG and SFB could be influenced by these factors but it has to be considered, that we are talking about very small differences, the maximum number of aphids was 4 per plant. Therefore, the information given by the second evaluation is rather limited.
It has to be considered that three replications are quite a low number, but the results for the PEB and SFB treatment in the first evaluation showed very small confidence intervals and the plots needed a minimum size on a limited field space to evaluate the infestation from the border.
If foil is already in use, the only potential cost-neutral plant protection measure known to the authors is the changing of colour. All other measures (i.e. nets, beneficials) have additional costs. Further studies of the contrast aspect in the colonization of crops by winged pests might show the potential of the measure, in combination with other integrated measures, as part of an integrated pest management system to minimize the need of intervening actions. The spray film used in the experiments will be developed further and tested in consecutive years and/or more than one field to better understand its capacity of deterring aphids.
The author(s) declare that they do not have any conflicts of interest.
Supplementary information (Figs. S1-S5; Tables S1-S18; the R-code for modelling; ANOVA tables; model-based means of the scaled rates and their corresponding comparisons) is available at https://github.com/MaxMenssen/niemann_et_al_2022.
Barro, P.J., 1991: Attractiveness of four colours of traps to creal aphids in south Australia (Hemiptera: Aphididae). Australian Journal of Entomology 30 (4), 263–264, DOI: 10.1111/j.1440-6055.1991.tb00431.x.
Bates, D., M. Mächler, B. Bolker, S. Walker, 2015: Fitting Linear Mixed-Effects Models Using lme4. Journal of Statistical Software 67 (1), 1–48, DOI: 10.18637/jss.v067.i01.
Blackman, R.L., V. F. Eastop, 2000: Aphids on the world's crops: An identification and information guide. New York, Wiley, 466 pp., ISBN: 0471851914.
Diaz, B.M., M. Muńiz, L. Barrios, A. Fereres, 2007: Temperature Thresholds and Thermal Requirements for Development of Nasonovia ribisnigri (Hemiptera: Aphididae). Environmental Entomology 36 (4), 681–688, DOI: 10.1603/0046-225x(2007)36[681:ttatrf]2.0.co;2.
Döring, T.F., 2014: How aphids find their host plants, and how they don't. Annals of Applied Biology 165 (1), 3–26, DOI: 10.1111/aab.12142.
Döring, T.F., L. Chittka, 2007: Visual ecology of aphids—a critical review on the role of colours in host finding. Arthropod-Plant Interactions 1 (1), 3–16, DOI: 10.1007/s11829-006-9000-1.
Döring, T.F., S.M. Kirchner, S. Kuhne, H. Saucke, 2004: Response of alate aphids to green targets on coloured backgrounds. Entomologia Experimentalis et Applicata 113 (1), 53–61, DOI: 10.1111/j.0013-8703.2004.00208.x.
Döring, T.F., K. Röhrig, 2016: Behavioural response of winged aphids to visual contrasts in the field. Annals of Applied Biology 168 (3), 421–434, DOI: 10.1111/aab.12273.
Döring, T.F., J. Spaethe, 2009: Messungen der Augengröße und Sehschärfe bei Blattläusen (Hemiptera: Aphididae). Entomologia Generalis 32 (2), 77–84, DOI: 10.1127/entom.gen/32/2009/77.
Farnier, K., A.G. Dyer, M.J. Steinbauer, 2014: Related but not alike: not all Hemiptera are attracted to yellow. Frontiers in Ecology and Evolution 2 (67), DOI: 10.3389/fevo.2014.00067.
Finch, S., R.H. Collier, 2000: Host-plant selection by insects – a theory based on 'appropriate/inappropriate landings' by pest insects of cruciferous plants. Entomologia Experimentalis et Applicata 96 (2), 91–102, DOI: 10.1046/j.1570-7458.2000.00684.x.
Greer, L., J.M. Dole, 2003: Aluminum Foil, Aluminium-painted, Plastic, and Degradable Mulches Increase Yields and Decrease Insectvectored Viral Diseases of Vegetables. HortTechnology 13 (2), 276–284, DOI: 10.21273/HORTTECH.13.2.0276.
Holman, J., 2009: Host Plant Catalog of Aphids: Palaearctic Region. Dordrecht, Springer Netherlands, DOI: 10.1007/978-1-4020-8286-3.
Hommes, M., G. Siekmann, O. Piepenbrock, U. Baur, A. Fricke, T. Thieme, 2003: Reduzierung des Blattlausbefalls an ausgewählten Gemüsekulturen durch Mulchen mit verschiedenen Materialien und Farben: Reducing aphid infestations in selected field vegetables with mulches of different materials and colours. URL: https://orgprints.org/16630/1/16630-02OE097-ble-jki-2003-blattlausbefall.pdf. Access: 14. July 2020.
Hooks, C.R.R., A. Fereres, 2006: Protecting crops from non-persistently aphid-transmitted viruses: a review on the use of barrier plants as a management tool. Virus research 120 (1-2), 1–16, DOI: 10.1016/j.virusres.2006.02.006.
Johnson, G.V., A. Bing, F.F. Smith, 1967: Reflective Surfaces Used to Repel Dispersing Aphids and Reduce Spread of Aphid-Borne Cucumber Mosaic Virus in Gladiolus Plantings. Journal of Economic Entomology 60 (1), 16–18, DOI: 10.1093/jee/60.1.16.
Jones, F.R., R.K. Chapman, 1968: Aluminum foil and other reflective surfaces to manipulate the movement of aphid vectors of plant viruses: Proceedings of the Entomological Society of America 23 (23), 146–148.
Kift, N.B., A. Mead, K. Reynolds, S. Sime, M.D. Barber, I. Denholm, G.M. Tatchell, 2004: The impact of insecticide resistance in the currant-lettuce aphid, Nasonovia ribisnigri, on pest management in lettuce. Agricultural and Forest Entomology 6 (4), 295–309, DOI: 10.1111/j.1461-9555.2004.00226.x.
Lenth, R., 2018: emmeans: Estimated Marginal Means, aka Least-Squares Means. R package version 1.2.3., URL: https://cran.r-project.org/package=emmeans. Access: 14. July 2020.
Moericke, V., 1950: Über das Farbsehen der Pfirsichblattlaus (Myzodes persicae Sulz.). Zeitschrift für Tierpsychologie (7), 263–274.
Moericke, V., 1955: Über die Lebensgewohnheiten der geflügelten Blattläuse (Aphidina) unter besonderer Berücksichtigung des Verhaltens beim Landen. Zeitschrift für Angewandte Entomologie 37 (1), 29–91, DOI: 10.1111/j.1439-0418.1955.tb00775.x.
Müller, H.J., 1964: Über die Anflugdichte von Aphiden auf farbige Salatpflanzen. Entomologia Experimentalis et Applicata 7 (1), 85–104, DOI: 10.1111/j.1570-7458.1964.tb00728.x.
Niemann, J.-U., M. Menssen, H.-M. Poehling, 2022: Reducing initial Aphid infestation by use of coloured mulch foils and newly developed biodegradable spray-films: raw data, statistical code and supplementary, URL: https://github.com/MaxMenssen/niemann_et_al_2022. Access: 9. February 2022.
R Core Team, 2019: R: A language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria, R Core Team.
Reinink, K., F.L. Dieleman, 1993: Survey of aphid species on lettuce. Bulletin OILB/SROP 16(5), 56–68.
Rufingier, C., L. Schoen, C. Martin, N. Pasteur, 1997: Resistance of Nasonovia ribisnigri (Homoptera: Aphididae) to Five Insecticides. Journal of Economic Entomology 90 (6), 1445–1449, DOI: 10.1093/jee/90.6.1445.
Shimoda, M., 2018: Recent advances in the optical control of insect pests using light and color: Special Publication of TARI No. 215, 87–102.
Straw, N.A., D.T. Williams, G. Green, 2011: Influence of sticky trap color and height above ground on capture of alate Elatobium abietinum (Hemiptera: Aphididae) in Sitka spruce plantations. Environmental Entomology 40 (1), 120–125, DOI: 10.1603/EN09344.
Wickham, H., 2017: tidyverse: Easily Install and Load the 'Tidyverse'. R package version 1.2.1., URL: https://cran.r-project.org/package=tidyvers. Access: 14. July 2020.