

Journal für Kulturpflanzen, 74 (07-08). S. 166–175, 2022 | DOI: 10.5073/JfK.2022.07-08.02 | Mair and Wolf
Monitoring of the development of honeybee colonies placed near apple orchards in South Tyrol during spring
Beobachtungen zur Volksentwicklung von Honigbienenvölkern im Einzugsgebiet des Südtiroler Obstanbaus während des Frühjahrs
 | (c) The author(s) 2022 This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/deed.en). |
Submitted/accepted for publication: 9 November 2021/1 July 2022 |
This monitoring provides data on colony development and mortality in front of free-flying honeybee colonies in the vicinity of South Tyrolian apple orchards during spring 2014–2016. We compared colonies in two areas, differing in the type of insecticides applied: AP-area with more treatments with active agents harmful for bees (Etofenprox and Chlorpyrifos-ethyl) than in the non-AP-area. According to the Liebefeld Method colony strength was evaluated five times every year from March–June, whereas the collection and counting of dead bees occurred parallel to this at least twice a week. Colonies placed in the AP-area did not show an increased bee mortality compared to colonies from non-AP-area. We found, however, a relationship between increased mortality and residues of harmful insecticides on the bees, independent of the area. Nevertheless, we found no differences in colony development between the two investigated areas, which could have been assignable to the different use of insecticides.
plant-protection-product-residues on dead honeybees, bee-harming insecticides, apple bloom
Diese Beobachtungen liefern Daten zur Volksentwicklung und zum Totenfall von freifliegenden Honigbienenvölkern im Einzugsgebiet des Südtiroler Obstbaus während des Frühjahrs. In diesem Monitoring überwachten wir Völker (AP-Gebiet und nicht AP-Gebiet), bei denen unterschiedliche Insektizideinsätze (mehr Einsätze im AP-Gebiet bei vergleichbaren Aufwandmengen für die Wirkstoffe Etofenprox und Chlorpyrifos-ethyl) in den Obstanlagen erfolgten. Die Volksstärken wurden mittels der Liebefelder Schätzmethode an fünf Terminen von März–Juni erhoben. Die Totenfallerhebungen hingegen erfolgten mindestens zweimal wöchentlich. Bei den Völkern aus dem AP-Gebiet (höherer Insektizideinsatz) konnten keine Unterschiede hinsichtlich der Häufigkeit oder Höhe der Mortalität im Vergleich zu den Völkern aus dem nicht AP-Gebiet festgestellt werden. An den Zeitpunkten, an denen eine deutliche Totenfallerhöhung festgestellt wurde, waren an Proben (aus beiden Gebieten in vergleichbarem Ausmaß) Rückstände von bienengefährlichen Pflanzenschutzmitteln festgestellt worden. In Bezug auf die Volksentwicklung konnten wir keine Unterschiede zwischen den Untersuchungsgebieten feststellen, welche auf den unterschiedlichen Insektizideinsatz zurückzuführen waren.
Pflanzenschutzmittelrückstände auf toten Honigbienen, bienengefährliche Insektizide, Apfelblüte
The most important agricultural production systems in South Tyrol are the cultivation of livestock and horticulture (Provincial institute for statistics, 2016). Horticulture is dominated by apple orchards and vineyards which are covering approximately 8% resp. 2.1% of the total useable farmland (Provincial institute for statistics, 2016). In addition to 5,600 ha of vineyards (more in the southern parts of the province), 115 ha of cherry orchards, 85 ha of apricots, 40 ha of pears and 165 ha of berries, such as strawberries, blueberries or raspberries (mostly grown at higher altitudes and heterogeneously distributed), by far the most produced fruit is apples on 18,312 ha, mainly in the Etschtal, Vinschgau and Eisacktal (Autonomous Province of South Tyrol, 2020). Most of the apple orchards are in the valley bottom but a few are growing on the hillsides of mountains of over 1,000 m a.s.l., between the prevailing meadows in these altitudes (Anese & Feichter, 2021). In general, in South Tyrol fields and farms have a relatively small structure with a median area per farm of about 2.55 ha (Dalla Via & Mantinger, 2012). Consequently, ownership and thus agricultural management varies distinctly within short distances. Both, a high density of farms and farmers on the arable land, as well as for honeybee colonies in South Tyrol a high density per km2 is true: South Tyrol holds more than 3,500 beekeepers with approximately 37,000 bee colonies, which results in a density of 5 bee colonies/km2 (Autonomous Province of South Tyrol, 2017; Südtiroler Imkerbund, 2021). This is quite high in comparison to 1.8 colonies/km2 in Germany and 2.4 colonies/km2 in Austria (Autonomous Province of South Tyrol, 2017). The whole year, 14,000–15,000 colonies are in the vicinity of apple orchards near the valley bottom and during bloom another 6,000-7,000 are brought to lower altitudes additionally by migrating beekeepers (Autonomous Province of South Tyrol, 2017).
The protection of bee colonies was always very important for farmers and beekeepers and therefore already in 1981 a provincial law was released to increase pollinators safety. In 2016, this law was updated and since prohibits the use of plant protection products harmful to bees1 during apple bloom (Autonomous Province of South Tyrol, 2016). This blooming period is well defined annually by official announcement for different altitude levels (for instance: 0-500 m a.s.l., 500-750 m a.s.l. and 750-1,000 m a.s.l.) to account for the later bloom of orchards in higher altitudes. The plant protection strategy varies every year and is adapted to different altitudes but also to the presence of diseases and pests. In particular, apple proliferation (AP), one of the most severe apple diseases, has spread in Burggrafenamt district (close to Meran) dramatically since 2006 (Österreicher & Unterthurner 2014a; 2014b). Therefore, farmers of this district applied a special strategy using insecticides to control Psyllids (Cacopsylla spp.) as the vector (explanation in Figure 1) of the disease. In the following, we will call this district "AP-area". Especially insecticides harmful to bees in spring (before and after the apple bloom) were used to stop a further spread of apple proliferation through the vectors Cacopsylla melanoneura and C. picta (Österreicher & Unterthurner, 2014a; 2014b; Fischnaller et al., 2019). The control of these vectors was, according to an increasing number of beekeepers, suspected to be the reason why their colonies suffered from abnormal behaviour in spring, such as increased mortalities or a weaker colony development (Wolf & Zelger, 2009). Treatments with plant protection products may contribute to pollinators decline (Potts et al., 2016). Chronic exposure to pesticides was associated with negative effects on bees (Sánchez-Bayo et al., 2016) as for example well described for the class of Neonicotinoids which affects foraging behaviour (Henry et al., 2012) or learning abilities (Decourtye et al., 2005) and can increase in combination with pathogen pressure individually honeybee mortality rates (Wallner, 1995; Alaux et al., 2010; Pistorius et al., 2021). In this study, we investigated the effects of the use of plant protection products harmful to bees before and after the apple bloom in South Tyrol on the development and the mortality of honeybee colonies. Therefore, we compared colonies in the AP-area and non-AP-area (low incidence of apple proliferation or nearly no presence), to examine if the indicators development and mortality differed between these two treatment zones. Additionally, no continuously recorded data on colony development or mortality of honeybee colonies in South Tyrol during spring is available, but collecting such data seemed to be necessary, in order to interpret better our observations.
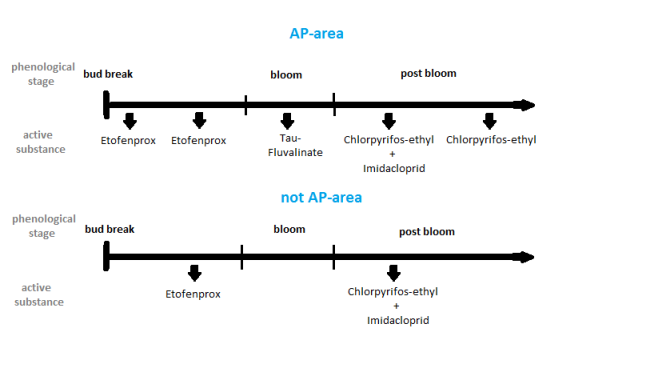
Fig. 1. Overview of the moments when different insecticides were used to limit the diffusion of the vectors of apple proliferation.
In 2014, when the monitoring started, on each of the 15 different sites 15 members from the South Tyrolean Beekeeping Association provided five colonies for a monitoring. Colonies were chosen randomly (no further specification on age of queen or dimension of colony requested) by the beekeepers and were separated from other colonies on the same apiary. New colonies were chosen year-wise; age of queens and overwintering success was not registered. Unfortunately, overwintering was not homogenous among the different apiaries: during the winter most colonies where not on the same apiary where they were investigated for our monitoring. In the years 2015 and 2016, due to some organizational issues, the number of sites had to be reduced to 13 (see Fig. S1). The monitored apiaries were distributed over the Etschtal from the area around the city Meran up to the area around the Research Centre Laimburg at Pfatten (see Table S2 and Fig. S1). The length of the observed valley section is about 40 km. About two thirds of the observed apiaries were in the AP-area (10 in 2014 and 9 in 2015 and 2016) and about one third (5 in 2014 and 4 in 2015 and 2016) in the non-AP-area. In the AP-area, more treatments were made with insecticides to limit the diffusion of the vectors (C. melanoneura and C. picta) of apple proliferation. The differences between both areas are shown by the recommended treatments with insecticides in Fig. 1: to protect against psyllids treatments with the active agents Chlorpyrifos-ethyl, Etofenprox and Tau-Fluvalinate, and to protect against Dysaphis plantaginea or Eriosoma lanigerum an Imidacloprid application (in case of necessity based on occurrence of pest) were made (strategy can be seen as exemplary for spring 2014–2016) (Österreicher & Unterthurner, 2014a; 2014b; Fischnaller et al., 2019; Südtiroler Beratungsring für Obst- und Weinbau, 2014; 2015; 2016).
Beekeepers were free with regards to the way in which they conducted colony management, which they integrated in the monitoring. Colonies were observed every year from the mid-March to mid-June. In each year, five assessments of colony-strength were made according to the Liebefeld Method every 21 days. The first one was performed at the middle-end of March and the last at the beginning-middle of June. Usually, all colonies of the 15 (in 2014) and 13 (in 2015 and 2016) sites, respectively, were evaluated within one calendar week (abbreviated in this article with cw, exact dates of evaluations in Table S3). In addition, the number of dead bees accumulating in front of the hives was registered. The sampling methods of bees differed amongst the years: The first year a tarp in front of the hives for collection was used (see Fig. S4), whereas in the 2nd and 3rd year on some sites underbaskets were used (only on 3 sites for all 5 colonies) (for details see Table S4). Generally, dead bees were counted and collected twice a week. If the point in time or the amount of an increase in mortality was interesting, a sample was taken and stored in a freezer for potential chemical residue analysis. Due to the two sampling methods (tarp or underbasket), it was not possible to determine a threshold-value for analysis. In addition, the limited budget for residue analysis was used to choose the most important increments of mortality of each apiary. The residue analyses were done mostly by the chemical laboratory at the Laimburg research centre. A few samples were analysed by the laboratories pH (Tavernelle Val di Pesa, Italy), Greit (Bologna, Italy) and the Julius Kühn Institute (JKI). Data were analysed and all figures created using the computer software R (R Core Team, 2016). The effect of the number of adult bees in the colonies at the mid/end of March (1st evaluation) on the number of adult bees in the colonies at the beginning/mid of June (5th evaluation) (in Fig. 5) were analysed using a linear model and a subsequent one-way ANOVA and a Tukey-test.
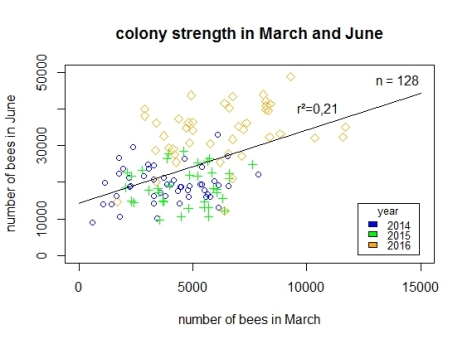
Fig. 5. Regression between the adult bee population in March at the start and that one in June at the end of the observations. The green crosses represent the data from 2014, the blue circles data from 2015, and the orange squares the data from 2016.
For the bee mortalities we show three figures (Fig. 2, Fig. 3 and Fig. 4). Figure 2 shows exemplarily the result of two apiaries (Tyrol from the AP-area and Kaltern from the not AP-area – on both dead bees were collected with a tarp) where in a more detailed way the exact number of dead bees observed is shown together with the results of some residue analysis. The observed number of dead bees shows different trends between the three years of monitoring and the two different sites. Every year the number of dead bees increased with ongoing observations (from March until June) and every year some clear increments were observed. For some peaks of mortality, residues of plant protection products harmful to bees where found (like at the end of May in Tyrol 2015 with 0,69 mg/kg Chlorpyrifos-ethyl).
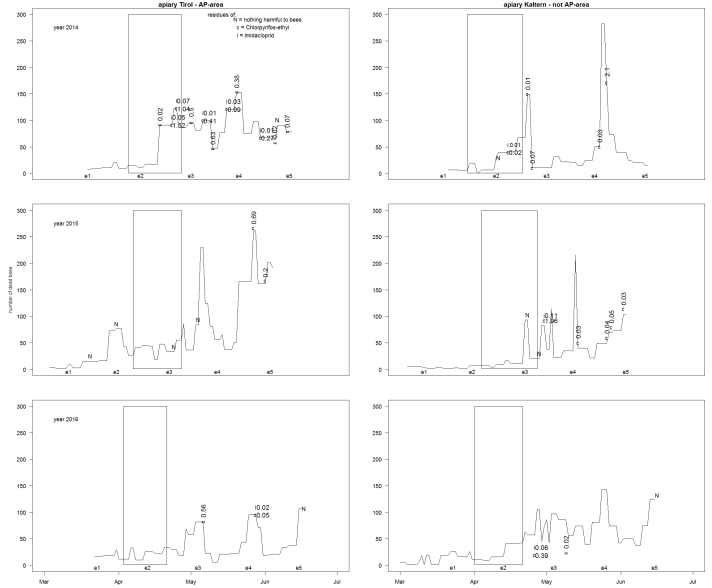
Fig. 2. Sum of dead bees per day registered in front of the hives of apiary Tyrol (left row) and apiary Kaltern (right row). The rectangles represent the periods of apple bloom. The times when colony-size was evaluated are also marked (for example e1 = evaluation of colony size no. 1 and marks the week in which the evaluations were made). The letters (N = nothing harmful to bees, c = Chlorpyrifos-ethyl, i = Imidacloprid) above or below the lines indicate the moment when dead bees were analysed for residues and whether insecticides harmful to bees were found on them or not.
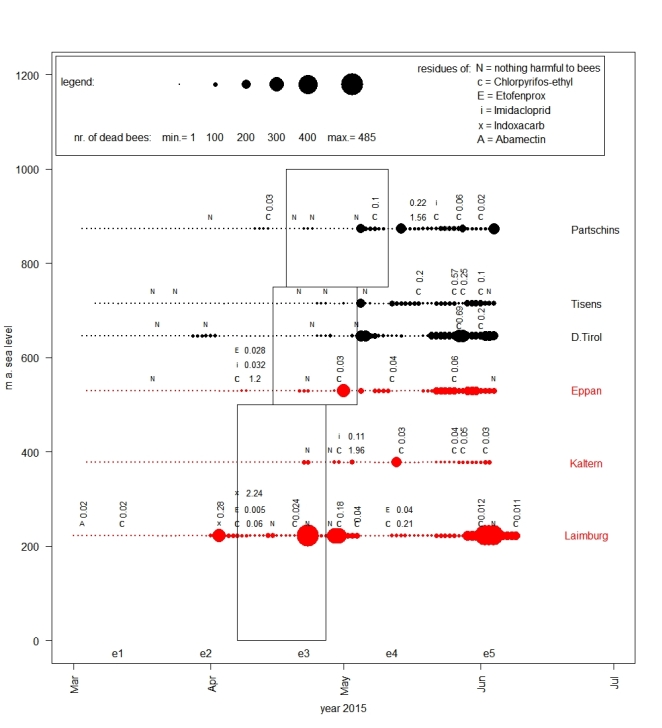
Fig. 3. Overview of the intensities of mortality on different apiaries during the observation in 2015. Points indicate the intensity of mortality (sum of the dead bees of 5 colonies per day) at the different apiaries. The bigger the circles, the more dead bees were found in front of the hives. In one line the results of one apiary are represented and at the end of the line its name is listed. For this figure only 3 representative apiaries from each of the two investigated areas (AP-area = black and non-AP-area = red) were chosen to avoid overlapping of data points and consequently reduced visibility of the results. The rectangles correspond to the periods of apple bloom according to the different altitudes. The times when colony-size was evaluated are also marked (for example e1 = evaluation of colony size no. 1 and marks the week in which all the evaluations were made). The letters (N = nothing harmful to bees, c = Chlorpyrifos-ethyl, E = Etofenprox, i = Imidacloprid, × = Indoxacarb and A = Abamectin) above the circles indicate whether samples of dead bees were contaminated by insecticides harmful to bees or not.
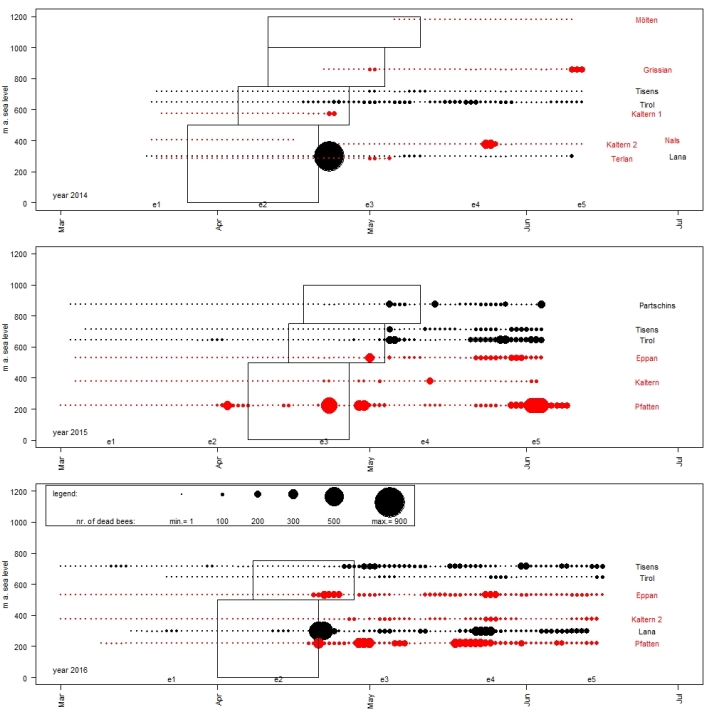
Fig. 4. Overview of the intensities of mortality on different apiaries from 2014–2016. Points indicate the intensity of mortality (sum of the dead bees of 5 colonies per day): the bigger the circles, the more dead bees were found in front of the hives. In one line the results of one apiary are represented and at the end of the line its name is listed. For this figure only 3 representative apiaries from each of the two investigated areas (AP-area = black and non-AP-area = red) were chosen for each year to avoid overlapping of data points and consequently reduced visibility of the results. The rectangles correspond to the periods of apple bloom according to the different altitudes. The times when colony-size was evaluated are also marked (for example e1 = evaluation of colony size no. 1 and marks the week in which all the evaluations were made).
Figure 3 is limited on the results of three apiaries of each of the two investigated areas from the year 2015 on different altitudes. The apple bloom is indicated by rectangles according to the provincial law no. 8 from 2016 for the different altitudinal levels 0-500 m a.s.l., 500-750 m a.s.l. and 750-1000 m a.s.l. Circle size indicates the intensity of mortality during the observation period and shows that most of the biggest circles were observed after apple bloom. Only a few enlarged circles were observed in the period before blooming: for example, at apiary D. Tyrol on April 2 with 77 and at apiary Pfatten on April 3 with 271 dead bees. At the last-mentioned apiary and the apiaries Partschins and Eppan increased mortalities were registered before the end of the bloom (Pfatten on April 23 with 485, Eppan on May 1 with 280 and Partschins on May 5 with 200 dead bees) – numbers which were never observed before the end of bloom on other apiaries (see Table S8). Immediately after the end of the bloom, at four of these in total six representative shown apiaries in Fig. 3 mortalities of more than approximately 200 bees were observed (Pfatten (318), Tirol (302), Tisens (194) and Partschins (200)). Similar mortalities were observed on apiary Kaltern on May 13 (216) and at the end of May on the apiaries D. Tyrol (maximum on May 20 and 21 with 293) and Eppan (maximum on May 29, 30 and 31 with 183). Other increased mortalities were observed at the beginning of June on the apiaries Pfatten (maximum on June 2, 3 and 4 with 445), Partschins (maximum on June 4 with 236) and D. Tyrol (maximum on June 2 and 3 with 221). Whether samples of dead bees were contaminated or not contaminated with active agents/insecticides harmful to bees, is indicated in our plot by letters.
Figure 4 gives an overview of the mortality registered in front of some apiaries for the three years of observation. Presented data are limited on the results of three apiaries each of the two investigated areas each year. Most of the strongest increases in mortality were observed after apple bloom. Before bloom, mortality was more or less constant with little variations/exceptions: for example, at apiary Tyrol and Pfatten in 2015 or in 2016 at the apiaries Tisens and Lana. At some apiaries increased mortalities were registered before the end of the bloom, for example, at three apiaries in 2014 (Kaltern 1, Tirol and Grissian) and three in 2015 (Pfatten, Eppan and Partschins) and at four in 2016 (Pfatten, Lana, Eppan and Tisens) (for exact numbers see Table S8). Every year, within a few days immediately after the end of the bloom-period, at most of this representatively shown apiaries mortality increased, like at apiary Lana in 2014 and 2016 or in Pfatten 2015 and 2016. Moreover, at some apiaries some occasional, momentary increased mortalities were observed, for example at the end of May (Lana 2016) or the beginning of June (Pfatten 2015).
For those points in time at which most of the dead bees in front of the hives of an apiary were registered, it was often possible to take a sample and analyse it later in the laboratory for chemical residues. In the 210 of the 347 analysed samples (overview in Table S7), in which products harmful to bees were found, Chlorpyrifos-ethyl was the most frequently detected substance with 206 findings. Imidacloprid and Indoxacarb were detected 62 and 12 times, respectively. The highest absolute concentration was measured for Chlorpyrifos-ethyl, with 32.83 mg/kg, and the second highest for Imidacloprid (1.49 mg/kg) (Table 1).
Table 1. Mean and maximum values as well as no. of detections for products harmful to bees found in the residue analyses on dead bees during the monitoring.
active substance | max [mg/kg] | mean [mg/kg] | st. dev. [mg/kg] | no. of detections |
Abamectin | 0.029 | 0.02 | 0.008 | 5 |
Chlorantraniliprole | 0.028 | 0.02 | 0.008 | 5 |
Chlorpyrifos-ethyl | 32.83 | 0.62 | 2.39 | 206 |
Chlorpyrifos-methyl | 0.18 | 0.1 | 0.12 | 2 |
Dimethoate | 0.55 | 0.12 | 0.18 | 9 |
Emamectin | 0.01 | 0.01 |
| 1 |
Etofenprox | 0.06 | 0.03 | 0.02 | 6 |
Imidacloprid | 1.49 | 0.12 | 0.24 | 62 |
Indoxacarb | 2.24 | 0.29 | 0.62 | 12 |
Spinosad | 0.09 | 0.08 | 0.01 | 2 |
Thiamethoxam | 0.02 | 0.02 | 0.003 | 2 |
From 2014–2016, bee colonies which at the end of March started with an adult population of more than 7,500 were 13 in total: one in 2014 and 2015 and eleven in 2016. The median for the adult population at the 1st evaluation was 3,312 in 2014, 4,856 in 2015, and 6,004 in 2016. These populations were all significantly different from each other (p-value always < 0.01). This clear difference especially from the year 2016 in comparison to the other two years is visible in Fig. 5: the orange squares (2016) are clearly shifted more to the right on the x-axis in comparison to the blue spots of 2014 or the green crosses from 2015. Furthermore, in the first year of monitoring, some bee colonies started with a very low number of adult bees. In 2014, six colonies had an adult population <= 1500 bees/colony, whereas in 2015, this was the case for only one colony and in 2016 for none. Looking at the distribution of the colony-strengths at the last evaluation of colony size in Fig. 5, it is visible, that colonies from 2016 are more in the upper third of the y-axis than those from 2014 and 2015. This difference in the median of adult population size is statistically significant (2014: 18,956, 2015: 18,283 and 2016: 34,430 bees; p-value < 0.01). In addition, the dimension of the adult population at the 1st evaluation of colony-size correlates significantly with the dimension of the adult population at the 5th determination (+ 0.47). The relationship between adult population at the 1st and the 5th evaluation of colony size is weak (lm in R showed a r2 of 0,21) but statistically significant (p-value < 0.01).
Figure 6 shows the number of bees per colony at time points of colony size evaluation for the two different analysed areas "AP-area" and "non-AP". Generally, boxes increased from the 1st until the 5th evaluation in both areas (except "non-AP" at the 4th evaluation), indicating the increase of variability of colony sizes. Only at the 2nd evaluation of colony size a significant difference between the no. of bees in the hives of the two different areas was observed, while at the other four evaluations, no differences were recorded.
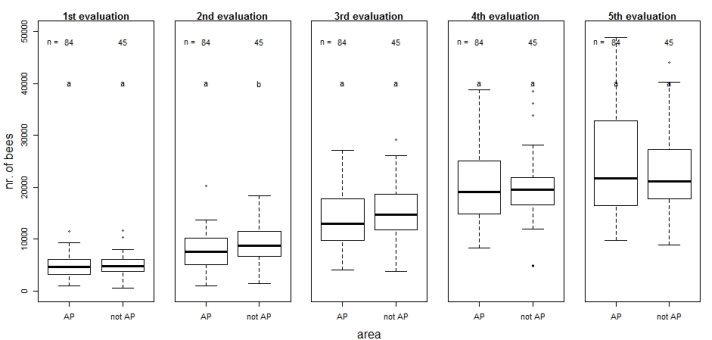
Fig. 6: Number of adult bees in the observed colonies from 2014–2016 in the two analysed areas ("AP"= high incidence of Apple Proliferation; "non-AP"= low incidence of Apple Proliferation) at the five points in time of colony size evaluation.
This is the first study, which provides detailed information on mortality in front of honeybee colonies and colony development in South Tyrol for spring. We observed more than 60 colonies on at least 13 different apiaries in the vicinity of apple orchards over three years in areas, where different strategies for the use of insecticides harmful to bees were followed. No differences were found, when comparing the accumulation of dead bees in front of the hives of the two different investigated areas. Unfortunately, no insecticide-free area for comparison was available. Even if the collection of dead bees was done in two different ways (tarp or underbasket trap) the tendency observed was always the same: moments of increments and numbers of dead bees registered in both areas (AP-area and non-AP-area) are comparable (see Figs. 2–4). Generally, numbers of collected dead bees increased from March until June. This is not surprising as considering the growing adult bee population in the colonies. Dead bees of some of the most intensive mortalities were analysed for plant protection product residues and in most of the cases products harmful to bees were found. Although an increased mortality could also be caused by pathological problems like a Nosema- (Galajda et al., 2021) or a ACPV-infection (De Miranda et al. 2010; Bakonyi et al. 2002) or being the result of the “Maikrankheit”, the residues of products harmful to bees detected, often reached very high concentrations which increases the probability that these were the cause for the increased mortality. At the moment of the highest mortality during the bloom at Pfatten in 2015 (April 23), no residues of plant protection products were found. Since it is such a strong increase, the colony evaluation, using the Liebefelder method, (what we also often identified as a cause for an increase of mortality) on April 22 is improbable to be the only reason for it, because the impact of the evaluation to our experience is of a much lower impact. In our opinion residue-free dead bees in a moment of a sudden increased mortality in front of the hives (despite a potential pathological origin as mentioned before) could also be product of an artefact, which has the following potential explanation: a) bees were contaminated with other chemical substances interfering with the residue analysis, and b) analysis laboratories were not specialized on bees as matrix with a low analysis sensitivity and consequently 22 of the 23 samples with no residues came from the same laboratory. Due to these inconsistencies among the different laboratories regarding their residue results we did not analyse further the detected concentrations but considered only presence/absence of substances. In addition, the number of dead bees and the detected residues on them will be an argument of another article, which is in preparation to be published. Unfortunately, no further analysis to check the presence of other pathogens were made. The two increased mortalities observed at the apiary Eppan at May 2 and at apiary Partschins on May 1 during the bloom in 2015 were exemplary for observations in the other two years. These mortalities were observed always immediately after the end of the bloom of the next lower altitudinal level. This shows that the foraging activity of honeybee colonies can overlap different altitudinal levels and cause increased mortalities during bloom. However, there are some strong indications, that some of the increased mortalities observed were caused at least in part by contact with plant protection products harmful to bees. First, time of intoxification (Fig. 2, Fig. 3 or Fig. 4) and the detected substances on many of the analysed samples of dead bees (Fig. 2 or Fig. 3) fit with the suggested applications (Fig. 1). Second, regularly when investigating the apiaries symptoms which indicate an intoxification, like bees laying on the back to the ground and spinning around or sticky dead/dying bees extracting their tongue or a high amount of dead/dying bees with pollen pellets on their legs were observed. Finally, also other publications of the same investigated area (Haller, 2017; Mair & Wolf, 2017; Ungerer, 2017; Mair, 2015) reinforce this assumption. The fact that we detected active agents before the treatment ban period (Chlorpyrifos-ethyl and Imidacloprid) suggests that some farmers applied these active agents contrary to the guidelines for pomiculture (Fig. 1). Whereas Chlorpyrifos-ethyl was used mostly to protect against the vectors of apple proliferation or the aphid Eriosoma lanigerum, Imidacloprid was used in apple orchards especially to protect against Dysaphis plantaginea. These investigations cannot clarify if the general applied strategy for products harmful to bees or a few inappropriate applications of the revealed highly harmful products are the reason for the observed intoxifications. On the one hand, the application of plant protection products harmful to bees follows a sincere and responsible approach (restrictions on etiquette of plant protection product, provincial law no. 15 from 2016 and information in the guidelines for pomiculture) and on the other hand, it is obviously impossible to completely avoid that honeybees are getting in contact with the applied substances. However, we cannot exclude other non-agricultural contamination sources, such as private gardens.
A general difference between the colony development on colonies in the AP-area and those from the non-AP-area could not be identified. The results are in line with the literature on bee development as described by Wille (1984), Bühlmann (1985), Liebig (1993a; 1993b), Imdorf & Maquelin (1993) or Imdorf et al. (2008). However, these researchers worked with harmonized colony strengths whereas we worked with a non-homogenous mix of colonies, which was provided by local beekeepers. In addition, we excluded from our valuations colonies which were very small, or when they produced queen cells or when they were used for building up new colonies (which must be strong colonies). Therefore, our approach tended probably to exclude the weakest and strongest colonies of the population.
The differences between the two areas in the adult population at the moment of the 2nd evaluation of colony-size (Fig. 6) can probably be explained by the fact, that only in the AP-area colonies were located at sites above 600 m a.s.l. and there in spring the season starts later than in the lower and climatically more mild sites below 600 m a.s.l. and consequently, this delays also the expansion of the colony dimensions. Generally, when comparing between the two analysed areas (AP-area vs. non-AP-area) we must consider that the AP-area-apiaries are located more in the northern part of South Tyrol than apiaries of the non-AP-area (see Fig. S1) with different climatical conditions. Finally, we have to consider that the samples of both areas are at least for 2015 and 2016 not equally balanced: 9 apiaries from the AP-area vs. 4 apiaries from the non-AP-area. It would have been better to increase sample-size for the non-AP-area but we had not enough resources in this project.
The study gives an overview of the observed effects of the use of products harmful to bees in apple orchards of South Tyrol before, during and after the bloom, on honeybee colonies placed in their vicinity. These data help to explain and understand better the real situation in the field and should offer the opportunity to discuss on an objective basis. Further investigations must examine why and when bees are coming into the orchards even after the end of the apple bloom to identify ways, how to increase protection for honeybees and other pollinators.
The article summarizes the results from the two projects Apistox and Controlbee conducted between 2014 and 2016. Sieglinde Mair and Hannes Schuler were also involved in the 2014 surveys. Both are thanked for their dedicated cooperation. In addition, a large part of the colonies of the sites surveyed during the three years had been provided and cared for by beekeepers of the South Tyrolean Beekeepers' Association. Without their cooperation and suggestions, the project could not have been carried out. Thanks are also due to the South Tyrolean Apple Consortium and to Department 31 of the South Tyrolean Provincial Administration, which co-financed the investigations.
The authors declare that they do not have any conflicts of interest.
The supplementary information for this article can be found online at https://doi.org/10.5073/JfK.2022.07-08.02
Alaux, C., J.-L. Brunet, C. Dussaubat, F. Mondet, S. Tchamitchan, M. Cousin, J. Brillard, A. Baldy, L. P. Belzunces, Y. Le Conte, 2010: Interactions between Nosema microspores and a neonicotinoid weaken honeybees (Apis mellifera). Environmental microbiology 12 (3), 774–782, DOI: 10.1111/j.1462-2920.2009.02123.x.
Anese, L., V. Feichter, 2021: Der Vinschger Apfel aus Südtirol. IDM Südtirol – Alto Adige, URL: https://www.vinschgau.net/de/essen-geniessen/lokale-produkte/apfel.html.
Autonomous Province of South Tyrol, 2016: Bestimmungen auf dem Gebiet des Pflanzenschutzes. Landesgesetz vom 15. April 2016, Nr. 8, URL: http://lexbrowser.provinz.bz.it/doc/de/204538%c2%a730/landesgesetz_vom_15_april_2016_nr_8/art_3_schutz_der_bienen_span_span.aspx.
Autonomous Province of South Tyrol, 2017: Neue Erkenntnisse zur Bienengesundheit. Über Zustand und Schutz der Bienenvölker in Südtirol wurde heute (6. Februar) bei einer Pressekonferenz informiert. Zugriff: 05.03.2021, URL: http://www.provinz.bz.it/news/de/news.asp?news_action=5&news_article_id=577820.
Autonomous Province of South Tyrol, 2020: Agrar- und Forstbericht 2020. Zugriff: 05.07.2022, URL: https://issuu.com/landsuedtirol-provinciabolzano/docs/forst-_agrar-katalog_deutsch_internet_2020?fr=sYjhiODczMDk3Nw.
Bakonyi, T., R. Farkas, A. Szendröi, M. Dobos-Kovács, and M. Rusvai, 2002: Detection of acute bee paralysis virus by RT-PCR in honey bee and Varroa destructor field samples: rapid screening of representative Hungarian apiaries. Apidologie, 33 (1) 63-74. DOI: https://doi.org/10.1051/apido:2001004.
Bühlmann, G. 1985: Assessing population dynamics in a honeybee colony. Mitteilungen der Deutschen Gesellschaft für Allgemeine und Angewandte Entomologie 4 (4-6), 312–316.
Dalla Via, J., H. Mantinger, 2012: Die Landwirtschaftliche Forschung im Obstbau Südtirols. Erwerbs-Obstbau 54, 83-115, DOI: 10.1007/s10341-012-0171-x.
Decourtye, A., J. Devillers, E. Genecque, K. Le Menach, H. Budzinski, S. Cluzeau, M.H. Pham-Delčgue, 2005: Comparative sublethal toxicity of nine pesticides on olfactory learning performances of the honeybee Apis mellifera. Archives of environmental contamination and toxicology 48 (2), 242–250, DOI: 10.1007/s00244-003-0262-7.
De Miranda, J.R., Cordoni, G., Budge, G., 2010: The Acute bee paralysis virus-Kashmir bee virus-Israeli acute paralysis virus complex. Invertebr Pathol. 103 (1) 30–47. DOI: 10.1016/j.jip.2009.06.014.
Fischnaller, S., M. Parth, M. Messner, E. Mitterrutzner, C. Mittelberger, R. Stocker, K. Janik, 2019: Monitoring psyllid vectors of apple proliferation in South Tyrol (Italy). A multi-year survey. Phytopathogenic Mollicutes 9 (1), 101-102, DOI: 10.5958/2249-4677.2019.00051.3.
Galajda, R., A. Valenčáková, M. Sučik, P. Kandráčová, 2021: Nosema Disease of European Honey Bees. Fungi 7 (9): 714. DOI: 10.3390/jof7090714.
Haller, M. 2017: A monitoring study to assess mortality and development effects on honeybee colonies placed in apple orchards of South Tyrol. Bachelor thesis. Free University of Bozen – Bolzano, Bozen. Naturwisschschaftliche Fakultät.
Henry, M., M. Béguin, F. Requier, O. Rollin, J.-F- Odoux, P. Aupinel, 2012: A common pesticide decreases foraging success and survival in honey bees. Science 336 (6079), 348–350, DOI: 10.1126/science.1215039.
Imdorf, A., C. Maquelin, 1993: Volksschätzung im zeitigen Frühjahr. Populationsmessung in Diskussion geraten. Schweizerische Bienen-Zeitung 116, 392–395.
Imdorf, A., K. Ruoff, P. Fluri, 2008: Volksentwicklung bei der Honigbiene. Forschungsanstalt Agroscope Liebefeld-Posieux ALP (Hrsg.). ALP forum 2008, Nr. 68 d. Zugriff: 13.11.2019, URL: https://www.agroscope.admin.ch/agroscope/de/home/themen/nutztiere/bienen/biologie/volksentwicklung/_jcr_content/par/columncontrols/items/0/column/externalcontent.bitexternalcontent.exturl.pdf/aHR0cHM6Ly9pcmEuYWdyb3Njb3BlLmNoL2RlLUNIL0FqYXgvRW/luemVscHVibGlrYXRpb24vRG93bmxvYWQ_ZWluemVscHVibGlr/YXRpb25JZD0yMDQ4Mw==.pdf.
Liebig, G., 1993a: Die biologischen Grundlagen der Volksentwicklung. Einführung in die Methode der Populationschätzung (I). Deutsches Bienen Journal 1993 (1), 14–15.
Liebig, G., 1993b: Die biologischen Grundlagen der Volksentwicklung (III) – Die Bruttätigkeit während der Saalweidenblüte. Deutsches Bienen Journal 1993 (3), 10–11.
Mair, B., 2015: Effetti dell'uso di prodotti fitosanitari pericolosi per le api: Risultati di un programma di monitoraggio in frutticoltura. Masterarbeit. Universitä degli studi di Padova, Padua. Dipartimento di Agronomia.
Mair, B., M. Wolf, 2017: Beobachtungen von Bienenvölkern im Südtiroler Apfelanbau. Obstbau Weinbau 54, (7/8), 29–34.
Österreicher, J., M. Unterthurner, 2014a: Starker Anstieg von Apfeltriebsucht im Burggrafenamt und Vinschgau. Obstbau Weinbau 51(2), 52–54.
Österreicher, J., M. Unterthurner, 2014b: Zusammenhänge zwischen Blattsaugerbekämpfung und Apfeltriebsuchtbefall. Vorschläge zur Abwehr 2014. Obstbau Weinbau 51 (2), 55–57.
Pistorius, J., D. Thorbahn, G. Bischoff, 2021: Jahresbericht der Untersuchungsstelle für Bienenvergiftungen für 2020. Journal für Kulturpflanzen 73 (3–4), 101-102, DOI: 10.5073/JfK.2021.03-04.06.
Potts, S.G., V. Imperatriz-Fonseca, H.T. Ngo, M.A. Aizen, J.C. Biesmeijer, T.D. Breeze, L. V. Dicks, L.A. Garibaldi, R. Hill, J. Settele, A.J. Vanbergen, 2016: Safeguarding pollinators and their values to human well-being. Nature 540 (7632), 220–229, DOI: 10.1038/nature20588.
Provincial institute for statistics, 2016: Landwirtschaft in Zahlen – Agricoltura in cifre. Autonomous Province of South Tyrol. Bozen. URL: https://astat.provinz.bz.it/de/aktuelles-publikationen-info.asp?news_action=300&news_image_id=899503.
R Core Team, 2016: R: A Language and Environment for Statistical Computing, Vienna, Austria. URL: https://www.R-project.org/.
Sánchez-Bayo, F., D. Goulson, F. Pennacchio, F. Nazzi, K. Goka, N. Desneux, 2016: Are bee diseases linked to pesticides? – A brief review. Environment international 89-90, 7–11. DOI: 10.1016/j.envint.2016.01.009.
Südtiroler Beratungsring für Obst- und Weinbau, 2014: Leitfaden Apfel. Lana: Pötzelberger Druck GmbH.
Südtiroler Beratungsring für Obst- und Weinbau, 2015: Leitfaden Apfel. Lana: Pötzelberger Druck GmbH.
Südtiroler Beratungsring für Obst- und Weinbau, 2016: Leitfaden Apfel. Lana: Pötzelberger Druck GmbH.
Südtiroler Imkerbund, 2021: Der Südtiroler Imkerbund und seine Struktur. Bozen. URL: https://www.suedtirolerimker.it/de/der-imkerbund/ueber-uns.
Ungerer, V., 2017: Vegetationskundliche Erhebungen des blühenden Unterwuchses in Südtirols Apfelanlagen und dessen Bedeutung für die Honigbiene. Bachelorarbeit. University of applied siences Weihenstephan-Triesdorf, München. Gartenbau und Lebensmitteltechologie.
Wallner, K., 1995: Bienenschäden im Weinbau. Versuche und Beobachtungen zu den Ursachen der Bienenschäden in Weinbaugebieten der Ortenau vor und während der Rebblüte. Stuttgart: Dr. Neinhaus Verlag AG.
Wille, H. 1984: Überlebensstrategien des Bienenvolkes. Liebefeld, 169–182. Zugriff: 10.12.2019, URL: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=1&ved=2ahUKEwik08b326rmAhXO26QKHf-GB9EQFjAAegQIBRAC&url=https%3A%2F%2Fwww.summ-summ.ch%2Fapp%2Fdownload%2F10847897595%2FWille_Ueberlebensstrategien.pdf%3Ft%3D1488210726&usg=AOvVaw03U6C3CBUR8JlbTvIBl0wT.
Wolf, M., R. Zelger, 2009: Bienenschäden während der Obstbaumblüte 2009. Obstbau Weinbau 47 (11), 375–377.
Footnotes:
In this article, products harmful to bees are those that are classified as harmful to bees according to the Italian etiquette. |